- 4192 Devonwood Way, Ashburn, Virginia, 20148
- Helpline: +1 (703) 665-3747
Glycobiology to Fight COVID-19
- Home
- Back to Journal
- Article Details
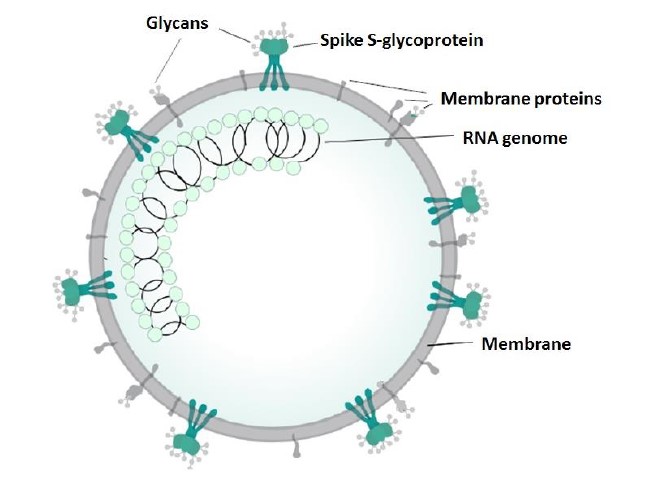
The current coronavirus COVID-19 (SARS-CoV-2) pandemics is an opportunity to look at the virus from a view of glycobiology, the field which deals with biological functions of carbohydrates, a synonym of the terms saccharides and sugars. Due to a structural complexity given by a larger number of functional groups, carbohydrates are less popular and generally also less known components of the living systems than nucleic acids and proteins. However, they are equaly important. Besides structural and nutritional role, carbohydrates attached to proteins or lipids function as signal, communication and cell traffic directing molecules. Metaphorically speaking, carbohydrates covalently bound to proteins, called glycans, serve as an address on a parcel or a name of the destination In this way glycans participate in initiation and regulation of a number of processes in living cells, but unfortunately, also in pathogenesis. This is also the case of viral infections, particularly by the group of glycosylated envelope viruses to which the new COVID-19 belongs. In coronaviruses all external domains of membrane-anchored proteins are glycosylated, and among them the most heavily glycosylated is the trimeric spike S-glycoprotein, which has the largest external domain (Figure 1).
Figure 1: Structure of a coronavirus. Modified from https://en.wikipedia.org/wiki/Coronavirus).
Major effort how to prevent infection by this extra dangerous virus is devoted to development of protective vaccines or monoclonal antibodies that would block the attachment of the virus to the human cell surface [1]. The main research target in this regard appears to be the viral spike S protein which was shown to associate with host membrane bound dipeptidyl peptidases [2-4]. There is a world-wide race for the first reliable and safe vaccine on the market. Before the availability of such a vaccine, the century-old convalescent plasma therapy, is still considered – the infusion of plasma rich in antibodies from survivors to sick persons [5].
I was quite surprised that the latest papers on COVID-19 do not pay attention to carbohydrates of both virus or host membrane proteins and their possible involvement in the infection process. The reason might be the insufficient knowledge of the pathogen due to its novelty. It has been shown that the primary receptors for several earlier identified coronaviruses are the host surface carbohydrates. MERS CoV binds to 9-carbon long acidic sugar sialic acid [6], while the human coronavirus HCoV-OC43 and bovine BoCoA virus recognize a modified sialic acid, which is acetylated at position 9, called 9-O-acetyl-sialic acid or 5,9-diacetyl-neuraminic acid [7]. In last two cases the diacetylated acidic sugar of the host is not recognized by the coronavirus S-glycoprotein, but by its hemagglutinin esterase (HE), also a transmembrane protein which has several domains. The first domain is responsible for the recognition of the 9-acetyl-sialic acid, the second domain is an esterase removing the 9-acetyl group thus modifying the receptor to sugar which is no longer recognized by another virus of the same type. The third HE domain is involved in the virus fusion with the host cell membrane. In two human coronaviruses a receptor for 9-O-acetyl-sialic acid binding occurs in one of the domains of the spike S-protein [8]. No information is available on a similar recognition of sugars by COVID-19 despite the presence of HE in the envelope [9].
In contrast to carbohydrate recognition, the first step suggested for COVID-19 entry into the cell is the attachment of the spike S-protein to one of the two host membrane peptidases occurring on the surface of lung, heart and kidney cells. The suggestion, originally based on a computer modeling of the spike S-protein binding to human peptidases ACE2 [2] and CD26 [3], was confirmed by cryo-electron microscopy structure of the complex of ACE2 with a receptor binding domain of the S-protein [4]. The function of ACE2 is to modify the angiotensin hormone regulating the blood pressure. The function of CD26 peptidase is signal transduction and regulation of the immune response.
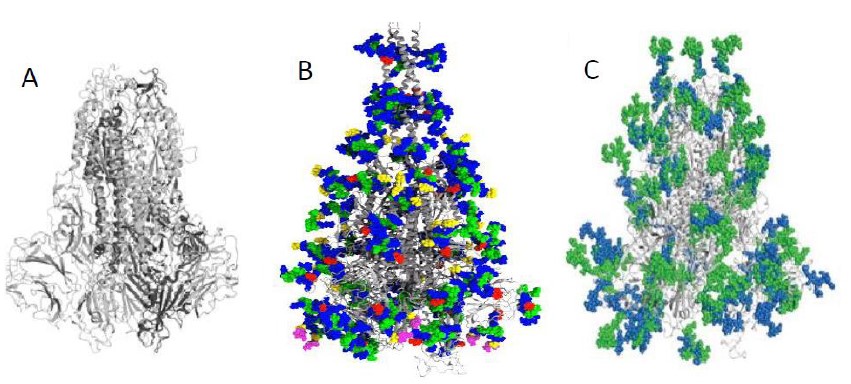
Both the spike S-protein and the two host receptor peptidases are heavily glycosylated, which means that they contain covalently linked oligosaccharide chains composed of 6 to 14 carbohydrate residues which are called glycans. The glycan attachment to protein requires a special amino acid sequence. Most of the glycans are built from residues of N-acetylglucosamine and mannose which are frequently further substituted by other sugars including sialic acid. The S-protein trimer of the SARS CoV-2 has more than 60 such glycosylation sites, by 16 more than its predecessor SARS-CoV [3] (Figure 2). The glycans cover the S-protein as a camouflage and protect the virus from the defense mechanisms of the host and proteolysis. The glycans are not uniform, they differ in chain length and terminal sugar residues (Figure 2), and the glycosylation pattern can vary even for a given glycosylation site. The glycans thus represent an extremely diverse array of immunochemical epitopes. No doubt that a potential preparation of the vaccine against a selected peptide portion of the S-protein covered in this way by complex carbohydrates requires a careful selection of the protein segment to be accessible to the corresponding antibodies in the glycan jungle. The extracellular domains of the transmembrane host peptidases ACE2 and CD26 suggested to serve as the primary targets for the COVID-19 spike S-protein are also glycosylated [11,12]. It is worth of noting that HE characterized in previous coronaviruses has 6 potential glycosylation sites [13].
Figure 2: Structure of the spike protein of COVID-19 without glycans (A) and with glycans (B, sugars are distinguished by color) and comparison of glycosylation of the spike proteins of COVID-19 (green) and SARS CoV (blue) (C). Modified from refs. 3 and 10.
The above considerations stress the importance of studies focused on the COVID-19 and host carbohydrate-protein interactions. Despite the evidence for binding of the spike S protein to ACE2 peptidase as the primary step of virus attachment [4], the possibility of earlier identified carbohydrate recognitions steps should not be dismissed. The glycans of the COVID-19 spike protein also represent an ideal opportunity to interact with host lectins on endothelial cells similarly to other coronaviruses SARS-CoV and HCoV-229E [14,15]. Detail knowledge of the structure of COVID-19 spike glycans as well as sugar specificity of its HE could lead to development of new therapeutics blocking the carbohydrate-protein interactions involved in the virus attachment and infection. Drugs based on carbohydrates could serve as antiadhesins on the surface of both virus and the cell. Interesting information supporting the glycobiology approach comes from the British company Iceny Diagnostics [16] developing a simple test for COVID-19 using an artificial receptor for the virus carbohydrates distinguishing influenza from coronavirus in saliva or nasal fluids with the aim to differentiate various coronaviruses. Antiviral drugs having high affinity for viral carbohydrate receptors could be developed based on the design and synthesis of sugar analogues, glycomimetics, and particularly of multivalent glycoclusters and glycodendrimers [17]. In summary, we could designate these strategies as carbohydrate or lectin directed anti-coronavirus drug design. Success of this approach will require a very close collaboration of carbohydrate organic chemists, structural biologists and virologists.
References
- Kruse RL (2020) Therapeutic strategies in an outbreak scenario to treat the novel coronavirus originating in Wuhan, China. F1000Res 9: 72. [View]
- Xu X. et al. (2020) Evolution of the novel coronavirus from the ongoing Wuhan outbreak and modeling of its spike protein for risk of human transmission. Science China Life Sciences 63: 457–460. [View]
- Vankadari N, Wilce JA (2020) Emerging COVID-19 coronavirus: glycan shield and structure prediction of spike glycoprotein and its interaction with human CD26. Emerging Microbes & Infections 9: 601-604. [View]
- Yan R. et al. (2020) Structural basis for the recognition of SARS-CoV-2 by full-length human ACE2. Science 367:1444-1448. [View]
- Westman N (2020) Why we’re still relying on a century-old strategy to treat COVID-19. The Verge. April 2020. [View]
- Li W et al. (2017) Identification of sialic acid-binding function for the Middle East respiratory syndrome coronavirus spike glycoprotein. Proc. Natl. Acad. Sci 114: E8508-E8517. [View]
- Schulze B, Herrler G (1993) Recognition of N-acetyl-9-O-acetylneuraminic acid by bovine coronavirus and hemagglutinating encephalomyelitis virus. Adv. Exp. Med. Biol. 342: 299-304. [View]
- Hulswit RJG et al. (2019) Human coronaviruses OC43 and HKU1 bind to 9-O-acetylated sialic acids via a conserved receptor-binding site in spike protein domain A. Proc. Natl. Acad. Sci. USA 116: 2681-2690. [View]
- Mousavizadeha L, Ghasemib S (2020) Genotype and phenotype of COVID-19: Their roles in pathogenesis. J Microbiol. Immunol. Infect. In press. [View]
- Baraniuk C (2020) Scientists scan for weaknesses in the SARS-CoV-2 spike protein. The Scientist, April 2020. [View]
- Prabakaran P et al. (2004) A model of the ACE2 structure and function as a SARS-CoV receptor. Biochem. Biophys. Res. Comm. 314: 235-241. [View]
- Aertgeerts K et al. (2004) Nâ€linked glycosylation of dipeptidyl peptidase IV (CD26): Effects on enzyme activity, homodimer formation, and adenosine deaminase binding. Protein Sci 13: 145–154. [View]
- Zeng Q et al. (2008) Structure of coronavirus hemagglutinin-esterase offers insight into corona and influenza virus evolution. Proc. Natl. Acad. Sci. USA 105: 9065–9069.
- Han DP, Lohani M, Cho MW (2007) Specific asparagine-linked glycosylation sites are critical for DC-SIGN- and L-SIGN-mediated severe acute respiratory syndrome coronavirus entry. J. Virol 81: 12029–12039. [View]
- Lozach PY, Burleigh L, Staropoli I, Amara A (2007) The C type lectins DC-SIGN and L-SIGN: receptors for viral glycoproteins. Methods Mol. Biol. 379: 51-68. [View]
- https://www.icenidiagnostics.com/news/
- Chabre ZM, Roy R (2012) Dendrimerâ€Coated Carbohydrate Residues as Drug Delivery Trojan Horses in Glycoscience. Pp. 407-438. In Dendrimer-based drug delivery systems: From theory to Practice. 1st Edition. Edited by Y.Cheng. 2012. John Wiley & Sons, Inc. [View]
Article Type
Opinion Article
Publication history
Received: April 27, 2020
Accepted: May 06, 2020
Published: May 09, 2020
Citation:
Biely P (2020) Glycobiology to Fight COVID-19. Med Case Rep Ther Stud 01(02): 10–12.
Peter Biely*
Institute of Chemistry, Slovak Academy of Sciences, Center of Glycomics, Bratislava, Slovakia
*Corresponding author
Peter Biely,
Institute of Chemistry,
Slovak Academy of Sciences,
Center of Glycomics,
Bratislava,
Slovakia;