- 4192 Devonwood Way, Ashburn, Virginia, 20148
- Helpline: +1 (703) 665-3747
COVID-19 and HIV Patients: A Case Report from S. Matteo University Hospital In Pavia (Italy)
- Home
- Back to Journal
- Article Details
Abstract
Coronavirus Disease 2019 (COVID-19) has been declared a pandemic since March 2020. Pneumonia caused by SARS-CoV-2 is associated with significant morbidity and mortality. The effect of HIV with low CD4 count on the course of COVID-19 is unknown. Here, we report successful recovery from SARS-CoV-2 pneumonia of a patient with HIV/AIDS, who was admitted to the Emergency Department of San Matteo University Hospital in Pavia. Furthermore, we did a brief review/comment of the related scientific literature. Apparently, HIV patients on therapy with normal lymphocyte are not at major risk than the general population.
Keywords: COVID-19, HIV, SARS-CoV-2, lung ultrasound.
Introduction
Starting in December 2019, an outbreak of a novel coronavirus (COVID-19) infection has been reported in Wuhan, China. The infection rapidly spread to other countries becoming a pandemic [1]. Responsible for this pandemic is a new type of Coronavirus, the seventh known in the world, classified as SARS-CoV-2. At the end of January 2020, the first imported cases of Coronavirus disease 19 (COVID-19) were discovered in Italy and a month after the COVID-19 epidemy outbroke in Italy.
Patients infected by the SARS-CoV-2 show symptoms like fever, cough, dyspnea, diarrhea, ageusia and anosmia, thoracic pain and usually present lymphocytopenia, thrombocytopenia, high levels of LDH, transaminases and/or creatine kinase. An interstitial pattern and subpleural lung consolidations are detected at thoracic ultrasound and ground-glass lung changes at Computed Tomography (CT) scan, in severe cases [2].
Older people, living with chronic diseases like diabetes, hypertension and/or chronic obstructive pulmonary disease, are more prone to severe COVID-19. Normally, people living with HIV with advanced disease, low CD4 count and high viral load and those who are not taking antiretroviral treatment, are at an increased risk of infections, but is unknown whether the immunosuppression of HIV will put a person at greater risk for COVID-19. Approximately 37,9 million people living with HIV [3] are at risk of infection with severe COVID-19. Cryptococcal, mycobacterial or pneumococcal pneumonia remain the commonest Human Immunodeficiency Virus (HIV)– associated complication in the antiretroviral therapy (ART) era. Anyway, there is still limited evidence on the role of viruses in HIV-associated pulmonary infections [4]. The first available data suggest that the host immune status, to some extent, may affect the outcomes of SARS-CoV-2 infection. However, there is lack of direct evidence for immune influence on the acute infection of SARS-CoV-2. Information on how SARS-CoV-2 infection impacts immunocompromised people, such as those living with HIV (PLWH), off therapy or on therapy, is of urgent concern to those managing PLWH. We know that during the SARS and MERS outbreaks, there were only a few case reports of mild disease among people living with HIV [5]. Up to now, there is no evidence that the risk of infection or complications of COVID-19 is higher among people living with HIV who are clinically and immunologically stable on antiretroviral treatment. Zhu and coworkers have described a single patient who recovered from the disease [6] while Guo and coworkers described 12 HIV/COVID-19 coinfected patients whose clinical phenotype was comparable to non-HIV patients [5]. However, in Europe, half of people with HIV have other chronic conditions that can increase the risk of an adverse outcome of COVID-19 infection.
Pneumonia caused by SARS-CoV-2 is associated with significant morbidity and mortality. The effect of HIV with low CD4 count on the course of COVID-19 is unknown. Here, we report successful recovery of one patient with HIV/AIDS from SARS-CoV-2 pneumonia. We describe in detail one case of HIV-positive patient admitted to our Emergency Department of San Matteo University Hospital in Pavia with a diagnosis of COVID-19 infection confirmed by viral test, and we briefly review the pertinent scientific literature.
Methods
According to our established protocol, every patient admitted to our Emergency Department with a suspect of SARS-CoV-2 infection was evaluated with a clinical visit, lung ultrasound, chest x-ray, needed blood tests, Arterial Blood Gas (ABG) analysis and Real time PCR (RT-PCR) for detection of coronavirus RNA on a nasopharyngeal swab. LUS (Lung US score) was derived from 12 ultrasound lung windows (2 anterior, 2 lateral, 2 posterior zones per hemithorax) and was attributed in function of A lines, B lines, regularity/irregularity of the pleural line and subpleural consolidations as previously described [7].
Results
Case report
On March 2020, a male, caucasian, 56 years old patient came to the attention of San Matteo Hospital Emergency Department in Pavia, presenting with fever and cough since 5 days, resistant to Amoxicillin/Clavulanic acid antibiotic therapy.
He was diagnosed with HIV in 1993. Currently in therapy with dolutegravir 50 mg Once Daily (OD) and lamivudine 300 mg OD, living with an HIV RNA viral load suppressed in a stable way since March 2007, when its nadir of CD4+ lymphocytes was 162 cell/ µl. On January 14, 2020 the HIV viral load was undetectable with 1878 total lymphocytes/µl and a CD4/CD8 ratio of 0.9.
As past medical history, he also presented major depression with suicide attempts, psychogenic headache, liver steatosis, neuropathy at lower limbs and previous infection of HBV (HbsAg negative, anti-HBsAg positive and anti-HBcAg positive). He was also taking the following daily drugs: bipyridine, delorazepam, gabapentin, haloperidol, mesoglilycan sodium and tamsulosin.
On admission, physical examination was normal, the body temperature was 38°C, with a systemic blood pressure of 115/75 mmHg, a cardiac rate of 86 bpm, a respiratory rate of 20 breaths per minute and the oxygen saturation was 95% while he was breathing ambient air. The results of ABG analysis, a Complete Blood Count (CBC) and Comprehensive Metabolic Panel (CMP) performed at admission are displayed in Table 1 and 2 respectively. To be noted, CPK and CPR were elevated, while platelet count was low.
Lung ultrasound showed a suggestive pattern of interstitial lung disease associated with bilateral subpleural consolidations in the postero-inferior scans (Figure 1, 2). The total Lung Ultrasound Score (LUS) was 10. Chest x-ray, performed at the admission, showed a mild thickening of the peribronchovascular interstitium with focal areas of consolidation in the right medio-superior and left medio-inferior regions, compatible with a viral interstitial pneumonia (Figure 3).
Table 1: Multi-day blood tests analysis of patient.
|
Blood Chemistry |
26-mar |
29-mar |
30-mar |
03-apr |
05-apr |
06-apr |
|
WBC (×103/ µl) |
4,1 |
4 |
|
4,2 |
3,77 |
|
|
Neutrophils (×103/µl) |
2,4 |
2 |
|
2,12 |
1,75 |
|
|
Lymphocytes (×103/µl) |
1,4 |
1,6 |
|
1,39 |
1,57 |
|
|
PLT (×103/µl) |
117 |
149 |
|
279 |
271 |
|
|
Hb (mmoL/L) |
9 |
8,38 |
|
7,94 |
8,19 |
|
|
CRP (mg/dl) |
5,09 |
8,86 |
|
2,13 |
0,92 |
|
|
LDH (mU/ml) |
296 |
359 |
|
|
|
|
|
Creatinine (µmol/L) |
93,3 |
86,63 |
|
90,17 |
93,7 |
|
|
Sugar (mmol/L) |
7,11 |
4,22 |
|
|
|
|
|
hsTnI (µg/L) |
4 |
|
|
<2,5 |
|
|
|
CPK (mU/L) |
341 |
1577 |
|
216 |
|
|
|
K+ (mEq/L) |
3,86 |
4 |
|
4,62 |
|
|
|
Na+ (mEq/L) |
135 |
141 |
|
138 |
|
|
|
Mg++ (mEq/L) |
2,2 |
|
|
|
|
|
|
Cl- (mEq/L) |
98 |
102 |
|
|
|
|
|
GPT (mU/ml) |
|
45 |
|
|
109 |
|
|
GOT (mU/ml) |
|
60 |
|
|
61 |
|
|
D-dimer (µg/L) |
|
|
|
3294 |
|
|
|
INR |
|
1,05 |
|
|
|
|
|
Fibrinogen (µmol/L) |
|
12,23 |
|
|
|
|
|
PCTI (µg/L) |
|
0,12 |
|
|
|
|
|
Ferritin (pmol/L) |
|
2867,17 |
|
2608,77 |
|
|
|
Albumin (g/dl) |
|
3,1 |
|
|
|
|
|
Interferon gamma (pg/ml) |
|
|
0,1 |
|
|
0,1 |
|
Interleukin-1 (pg/ml) |
|
|
176,12 |
|
|
0,1 |
|
Interleukin-10 (pg/ml) |
|
|
38,8 |
|
|
0,38 |
|
Interleukin-2 (pg/ml) |
|
|
0,1 |
|
|
10,41 |
|
Interleukin-6 (pg/ml) |
173,96 |
9,53 |
|
Interleukin-8 (pg/ml) |
534,5 |
44,81 |
|
TNF alpha (pg/ml) |
35,38 |
0,1 |
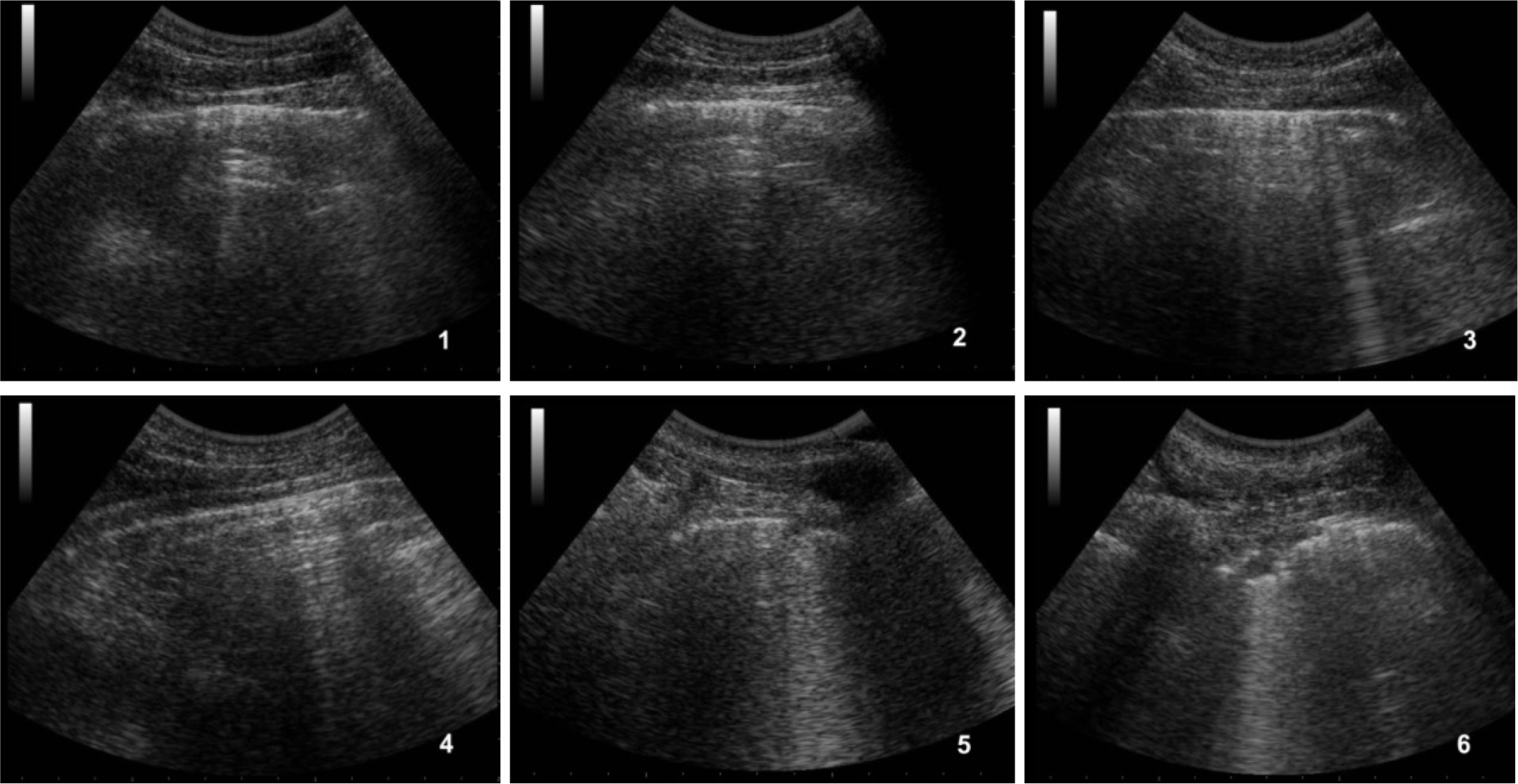
Figure 1: Ultrasound of the right lung.
1) antero-superior field with A-lines, 2) antero-inferior field with A-lines, 3) lateral-superior field with A-lines and few B-lines, 4) lateral-inferior field with A-lines, 5) postero-superior field with a small subpleural consolidation and B-lines, 6) postero-inferior field with subpleural consolidation.
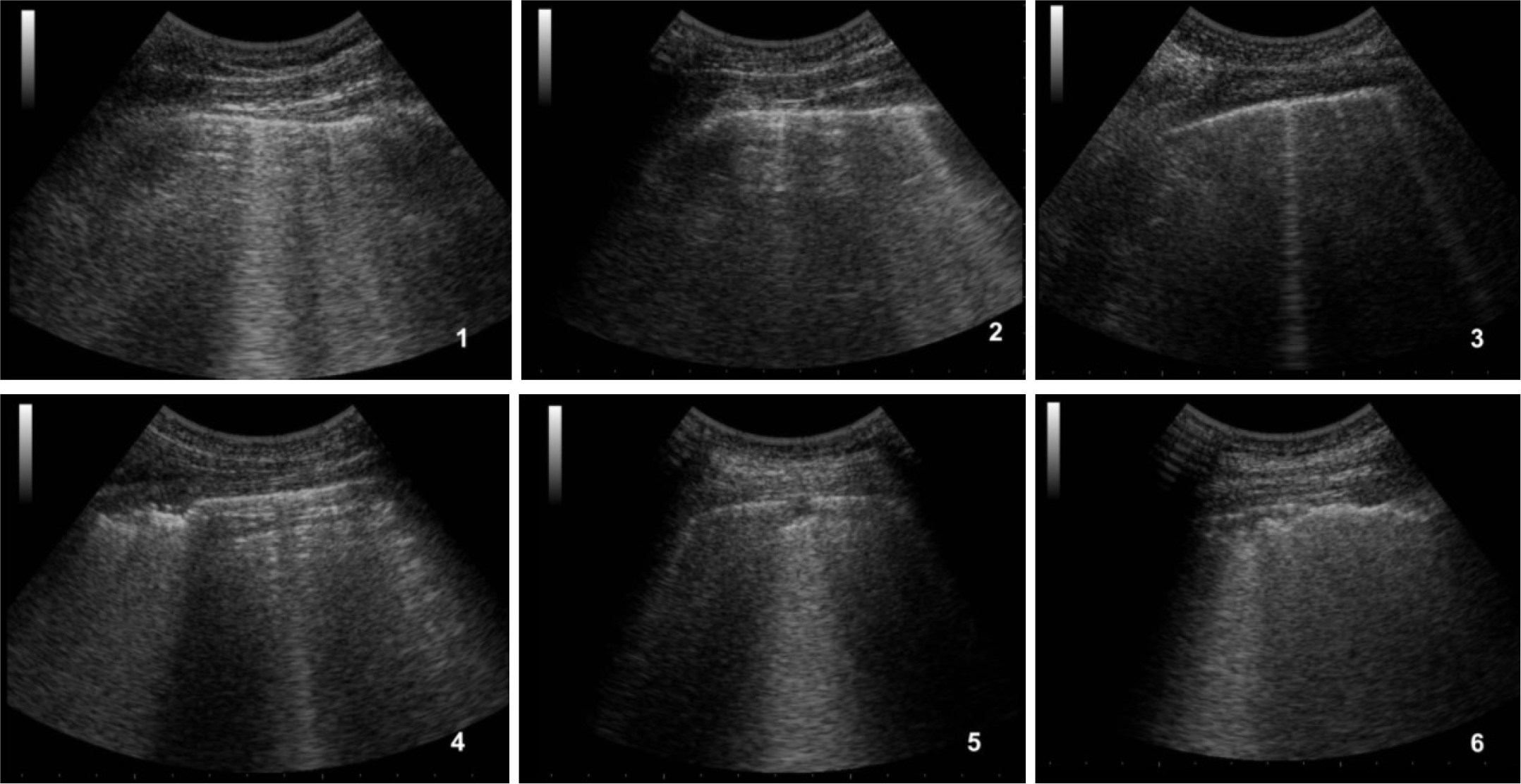
Figure 2: Ultrasound of the left lung.
1) antero-superior field with A-lines associated to some B-lines, 2) antero-inferior field with some coalescent B-lines, 3) lateral-superior field with two small B-lines, 4) lateral-inferior field with subpleural consolidations, 5) postero-superior field with a small subpleural consolidation, 6) postero-inferior field with confluent subpleural consolidations associated to fragmented pleural line.
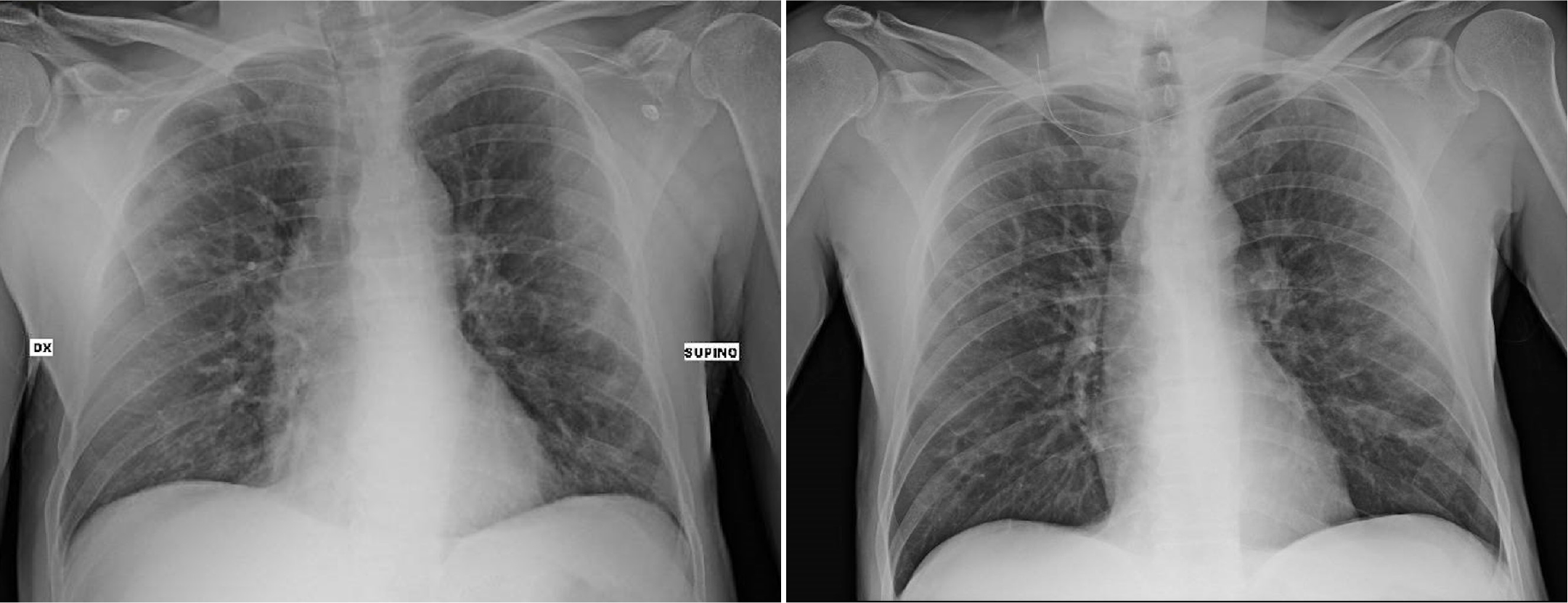
Figure 3: Chest x-ray at the admission (left) compared to that at the hospital discharge day (right).
Table 2: Arterial Blood Gas analysis of patient.
|
ABG |
value |
|
pH |
7,44 |
|
pCO2 (mmHg) |
35,8 |
|
pO2 (mmHg) |
63,8 |
|
FiO2 (%) |
21 |
|
SO2 (%) |
93,1 |
|
P/F (mmHg) |
304 |
|
Lac (mmol/L) |
1,5 |
|
HCO3– (mmol/L) |
24,2 |
|
GA (mmol/L) |
15,5 |
|
T (C°) |
38°C |
RT-PCR for detection of coronavirus RNA, performed on his nasopharyngeal swab on March 26, was weakly positive. Due to his symptoms, chest X-ray, LUS and his previous history associated with a swab positive for SARS-CoV-2, the patient was hospitalized in the Infectious Disease Department, become a COVID-19 ward. [8]
During the hospitalization, from March 26 to April 8, the patient was treated with hydroxychloroquine 200 mg every 8 hours, azithromycin 500 mg OD, ceftriaxone 1g OD, antithrombotic prophylaxis with heparin and hydration . He never needed O2 therapy.
On March 27 a second nasopharyngeal swab was performed which was clearly positive for SARS- CoV-2. The two following nasopharyngeal swabs performed on March 31 and April 4 were negative. He repeated blood test (showed in Table 1), with normalization of platelets and lymphocytes and decrease of CPR and CPK. Ferritin and D-dimer peaked at 2867,17 and 3294 respectively. Plasma levels of Interleukins 1–2-6–8-10, interferon gamma and TNF alpha were initially elevated, with a subsequent normalization of all levels, except for IL-6 and IL-8 that were greatly reduced. HIV blood tests were: HIV RNA (March 28) analysis was positive with a value of 225 cp/ml, total lymphocytes 1465/µl, CD4/CD8 1. He was also tested for Legionella and Streptococcus urinary antigens that resulted negative.
On April 7 a second chest x-ray was performed, showing a mild thickening of the peribronchovascular interstitium in the left middle-inferior field. (Fig 3). He was dismissed on April 8 in good clinical conditions, with an indication of a follow-up visit for HIV control.
Discussion
Two months after the outbreak of the epidemy in Italy there are a lot of questions not yet solved: are HIV people more or less resistant to COVID-19 infection? Do HIV patients with normal CD4 count on antiretroviral therapy behave differently from those with low CD4 count not taking therapy? Could patients with antiretroviral therapy be more protected from COVID-19? Moreover, if the clinical lung phenotype in severe cases is related to an exaggerated immune response, could the partially compromised immunity be the reason why HIV/AIDS patients do not show inflammatory changes and clinical symptoms? Or it is still possible that subtle alterations in the immune state of these patients could limit the HIV-infected patient response to the SARS-CoV-2 virus?
As detailed above, our patient developed a COVID-19 dependent interstitial lung syndrome, documented by lung ultrasound and chest x-ray upon arrival in the Emergency Department. As expected, blood test pattern was typical for COVID-19, such as lymphocytopenia (<1.5 x103/µl), CPR elevation, platelet reduction and increased levels of LDH. The patient showed good respiratory exchanges with a P/F > 300, but he needed a few days of hospitalization. The clinical presentation of this patient with SARS-CoV-2 pneumonia was similar to previously reported non-HIV infected cohorts in terms of symptoms, laboratory and radiological abnormalities. Notably, he showed lung involvement but did not progress to lung failure. Then, according to the biohumoral and radiological exams and on the basis of the prognosis of our patient, we can affirm that the clinical course was similar to that of non-HIV patients. This conclusion is in keeping with a recent meta- analysis on 70 patients by Cooper and coworkers, who concluded that well-controlled HIV patients are not at risk of poorer COVID-19 outcomes than the general population [9]. Mirzael and coworkers and Geretti and coworkers did find a slight higher mortality in co-infected HIV/COVID- 19 patients, probably due to older age and multimorbidity, not particular to HIV [10, 11].
Little is known regarding the severity of viral respiratory disease in HIV-infected individuals. It may be anticipated that the immune deficiencies associated with HIV infection may result in more severe illnesses due to respiratory viruses. Immunologic mechanisms, particularly cellular immune responses, are important components of the inflammatory response to respiratory viruses, so predicting illness severity in HIV-infected individuals becomes complicated.
Moderate to severe illnesses caused by these viruses in HIV-infected adults and children have been reported. Few prospective cohort or population-based studies are available with which to address their frequency. Also, it must be recognised that severe and even fatal respiratory illnesses can occur in the general population, albeit infrequently [12]. Humoral factors, and particularly serum antibody, are associated with at least partial protection from respiratory viruses in humans [13]. Pre- existing antibodies are associated with attenuated illness, although protection from subsequent re- infection may be incomplete. For example, children can be repeatedly infected with similar viruses, although subsequent illnesses are usually less severe than the primary infection [14]. Complete recovery from and clearance of these viral infections are likely mediated by virus-specific cellular mechanisms [15, 16].
HIV-infected individuals have deficiencies in both humoral and cellular defenses. Therefore, infection by common respiratory viruses may present differently in this population than in immunocompetent individuals. Since most adults with HIV disease acquired their infection as adults or adolescents, they probably have experienced infections by most common respiratory viruses and thus have some pre-existing immunity. On the basis of our case report, it is not possible to conclude whether the good prognosis of these patients was related or not to HIV condition and/or to the antiretroviral therapy, but in literature there are some evidence suggesting that in severe COVID-19 illness pathogenesis is, at least in part, supported by a cytokine storm syndrome, characterized by a fulminant hypercytokinaemia with multiorgan failure caused by the hyperactivation of the immune system. This syndrome is usually caused by an exaggerated response to viral infections and occurs in 3.7–4.3% of sepsis cases [17]. In this regard, it could be possible that a subtle suppression of the immune system, still associated to a clinically normal picture, is linked to the good prognosis of these patients.
If this assumption was true, then immunosuppression could be beneficial in the treatment of COVID-19, as suggested by several authors [18]. In this regard, it has been proposed that the immune alterations present in HIV patients could be protective from cytokine storm [5]. However, it must be kept in mind that, in HIV patients successfully treated with antiretroviral drugs, the immune system is usually normal, although subtle defects cannot be ruled out.
Moreover, it has been demonstrated that Coronaviruses, like HIV, are able to infect T lymphocytes, but the relative protective role of a co-infection is yet to be determined [19].
On the other hand, the opposite view that immunosuppressant drugs could impair a prompt and durable immune response of the organism, has also been raised [18]. Hence, the beneficial effects of reducing inflammation should be carefully weighed against the potential deleterious impairment of anti-microbial immunity. It is also possible that the antiretroviral HIV treatment by itself could in some way reduce the viral spread of SARS-CoV-2.
Furthermore, the duration of this new betacoronavirus shedding in HIV-infected adults is unknown and could affect quarantine and treatment recommendations. Despite relatively few published reports, respiratory viral infections in HIV-infected individuals appear to be prolonged compared with such infection in immunocompetent individuals. Impaired cell-mediated clearance of respiratory viruses from HIV-infected individuals may be responsible for these prolonged infections. Long-term shedding of respiratory viruses has important infection control implications, because HIV-infected individuals frequently visit medical facilities or are admitted to the hospital. In addition, prolonged respiratory viral shedding by HIV-infected individuals may have an impact on nursing homes, day care centers, or group foster homes, where other high-risk individuals may be exposed to these respiratory viruses [12]. However in our case report, after the first positivity, two consecutive nasopharyngeal swabs were negative since March 31, only 5 days after the first RT-PCR and 10 days after the symptoms insurgence.
Conclusion
In conclusion, duration of SARS-CoV-2 shedding in HIV-infected adults deserves evaluation in comparative studies. Future research should be directed to address the effect of HIV positivity on the clinical course of SARS-CoV-2 infections in larger cohorts, particularly in relation to different CD4 counts.
Abbreviations
COVID-19: Coronavirus Disease 2019;
SARS-CoV-2: Severe Acute Respiratory Syndrome - Coronavirus - 2;
LDH: lactate dehydrogenase; CT scan: computed tomography scan;
LUS: ultrasound lung score;
CBC: complete blood count;
CMP: comprehensive metabolic panel;
RT- PCR: real time polymerase chain reaction;
CPR: C Reactive Protein;
CPK: Creatine Phosphokinase;
CD4/CD8: ratio of CD4 to CD8 positive lymphocytes;
IL-1,2,6,8,10: interleukin 1,2,6,8,10;
TNF alpha: tumor necrosis factor alpha;
WBC: white blood cells;
PLT: platelets;
OD: once daily;
ABG: Arterial Blood Gas;
pCO2: partial pressure of carbon dioxide;
PO2: partial pressure of oxygen;
FiO2: fraction of inspired oxygen;
SO2: oxygen saturation;
LAC: lactates;
HCO3: bicarbonates;
AG: anion gap;
T: temperature.
Acknowledgment
We thank all the staff of the San Matteo Hospital who was deeply involved in the treatment of this pandemia in Lombardia: COVID-19 IRCCS San Matteo Pavia Task Force.
Infective Disease Unit: Staff: Raffaele Bruno, Mario U Mondelli, Enrico Brunetti, Angela Di Matteo, Elena Seminari, Laura Maiocchi, Valentina Zuccaro, Layla Pagnucco, Bianca Mariani, Serena Ludovisi, Raffaella Lissandrin, Aldo Parisi, Paolo Sacchi, Savino FA Patruno, Giuseppe Michelone, Roberto Gulminetti, Domenico Zanaboni, Stefano Novati, Renato Maserati, Paolo Orsolini, Marco Vecchia. Residents: Marco Sciarra, Erika Asperges, Marta Colaneri, Alessandro Di Filippo, Margherita Sambo, Simona Biscarini, Matteo Lupi, Silvia Roda, Teresa Chiara Pieri, Ilaria Gallazzi, Michele Sachs, Pietro Valsecchi.
Emergency Care Unit: Staff: Stefano Perlini, Claudia Alfano, Marco Bonzano, Federica Briganti, Giuseppe Crescenzi, Anna Giulia Falchi, Roberta Guarnone, Barbara Guglielmana, Elena Maggi, Ilaria Martino, Pietro Pettenazza, Serena Pioli di Marco, Federica Quaglia, Anna Sabena, Francesco Salinaro, Francesco Speciale, Ilaria Zunino; Residents: Marzia De Lorenzo, Gianmarco Secco, Lorenzo Dimitry, Giovanni Cappa, Igor Maisak, Giulia Maria Vezzoni, Benedetta Chiodi, Massimiliano Sciarrini, Bruno Barcella, Flavia Resta, Luca Moroni, Lorenzo Scattaglia, Elisa Boscolo, Caterina Zattera, Tassi Michele Fidel, Capozza Vincenzo, Marco Bazzini, Giulia Sturniolo, Elena Lago, Federico Bracchi.
Intensive Care Unit: Giorgio Iotti, Francesco Mojoli, Mirko Belliato, Luciano Perotti, Silvia Mongodi, Guido Tavazzi
Paediatric Unit: Gianluigi Marseglia, Amelia Licari, Ilaria Brambilla
Virology Unit: Staff: Barbarini Daniela, Bruno Antonella, Cambieri Patrizia, Campanini Giulia, Comolli Giuditta, Corbella Marta, Daturi Rossana, Furione Milena, Mariani Bianca, Maserati Roberta, Monzillo Enza, Paolucci, Stefania, Parea Maurizio, Percivalle Elena, Piralla Antonio, Rovida Francesca, Sarasini Antonella, Zavattoni Maurizio. Technical staff: Adzasehoun Guy, Bellotti Laura, Cabano Ermanna, Casali Giuliana, Dossena Luca, Frisco Gabriella, Garbagnoli Gabriella, Girello Alessia, Landini Viviana, Lucchelli Claudia, Maliardi Valentina, Pezzaia Simona, Premoli Marta. Residents: Bonetti Alice, Caneva Giacomo, Cassaniti Irene, Corcione Alfonso, Di Martino Raffella, Di Napoli Annapia, Ferrari Alessandro, Ferrari Guglielmo, Fiorina Loretta, Giardina Federica, Mercato Alessandra, Novazzi Federica, Ratano Giacomo, Rossi Beatrice, Sciabica Irene Maria, Tallarita Monica, Vecchio Nepita Edoardo.
Research Laboratories, Division of Infectious Diseases and Immunology: Antonella Cerino, Stefania Varchetta, Barbara Oliviero, Stefania Mantovani, Dalila Mele.
Pharmacy Unit: Monica Calvi, Michela Tizzoni.
Hospital Management: Carlo Nicora, Antonio Triarico, Vincenzo Petronella, Carlo Marena, Alba Muzzi, Paolo Lago.
Data Unit: Marco Rettani, Francesco Comandatore, Gherard Bissignandi, Stefano Gaiarsa, Claudio Bandi, Alessanda Ferrari , Francesca Pasi.
References
- World HealthOrganization (2020) Novel Coronavirus 2019 Events. [View]
- Guan WJ, Ni Z, Hu Y, et al. (2020) Clinical Characteristics of Coronavirus Disease 2019 in China. The New England journal of medicine 382: 18. [View]
- Joint United Nations Programme on HIV/AIDS (2020) Global HIV & AIDS statistics — 2020 fact sheet. UNAIDS . [View]
- Benito N, Moreno A, Miro JM, Torres A (2012) Pulmonary infections in HIV-infected patients: an update in the 21st century. Eur Respir J 39: 730–45. [View]
- Guo W, Fangzhao M, Yu D, Qian Z, Xiaoxia Z, et al. (2020) A Survey for COVID-19 Among HIV/AIDS Patients in Two Districts of Wuhan, China. SSRN Electron J 19. [View]
- Zhu F, Cao Y, Xu S, Zhou M (2020) Co-infection of SARS-CoV-2 and HIV in a patient in Wuhan city, China. J Med Virol 92: 529–530. [View]
- Di Pietro S, Mascolo M, Falaschi F, Brambilla W, Ruzga R, et al. (2020) Lung-ultrasound objective structured assessment of technical skills (LUS- OSAUS): utility in the assessment of lung-ultrasound trained medical undergraduates. J Ultrasound 8 : 1–9. [View]
- Asperges E, Novati S, Muzzi A, Biscarini S, Sciarra M, et al. (2020) Rapid response to COVID-19 outbreak in Northern Italy: how to convert a classic infectious disease ward into a COVID-19 response Centre. The J of Hospital Infection 105: 477–479. [View]
- Mirzaei H, Willi McFarland W, Mohammad Karamouzian M, Sharifi H (2020) COVID-19 Among People Living with HIV: A Systematic Review. AIDS Behav 30 : 1–8. [View]
- Cooper TJ, Woodward BL, Alom S, Harky A (2020) Coronavirus disease 2019 (COVID-19) outcomes in HIV/AIDS patients: a systematic review. HIV Med 2020: 1. [View]
- Anna Maria Geretti AM, Alexander Stockdale, Sophie Kelly, Muge Cevik, eSimon Collins, et al. (2020) Outcomes of COVID-19 related hospitalisation among people with HIV in the ISARIC WHO Clinical Characterisation Protocol UK Protocol: prospective observational study (preprint). [View]
- King J.C. Jr (1997) Community Respiratory Viruses in Individuals with Human Immunodeficiency Virus Infection. The American Journal of Medicine 102: 19–24 [View]
- Lamprecht CL, Krause HE, Mufson MA (1976) Role of maternal antibody in pneumonia and bronchiolitis due to respiratory syncytial virus. J Infect Dis 134: 211–217. [View]
- Glezen WP, Taber LH, Frank AL, Kasel JA (1986) Risk of primary infection and reinfection with respiratory syncytial virus. Am J Dis Child 140: 543–546. [View]
- McMichael AJ, Gotch FM, Noble GR, Beare PA (1983) Cytotoxic T cell immunity to influenza. N Engl J Med 309: 13–17. [View]
- Young DF, Randelli RE, Hoyle JA, Souberbielle BE (1990) Clearance of a persistent paramyxovirus infection is mediated by cellular immune responses but not by serum- neutralizing antibody. J Virol 64: 5403–5411. [View]
- Mehta P, McAuley DF, Brown M, Sanchez E, Tattersall RS, et al. (2020) COVID-19: consider cytokine storm syndromes and immunosuppression. The Lancet 395: 1033–1034. [View]
- Ritchie AL, Singanayagam A (2020) Immunosuppression for hyperinflammation in COVID-19: a double- edged sword?. The Lancet 395: 1111. [View]
- Wang X, Wei X, Gaowei H, Shuai X, Zhiping S, et al. (2020) SARS-CoV-2 infects T lymphocytes through its spike protein-mediated membrane fusion. Cell Mol Immunol 7 : 1–3. [View]
Article Type
Case Report
Publication history
Received: September 11, 2020
Accepted: September 18, 2020
Published: September 21, 2020
Citation:
Vezzoni GM, Barcella B, Vecchia M, Cappa G, Delorenzo M, et al. (2020) Covid-19 and HIV Patients: A Case Report from S. Matteo University Hospital In Pavia (Italy). Clar J Infect Dis Ther 01(02): 33–41.
Giulia M. Vezzoni1, Bruno Barcella1, Marco Vecchia2, Giovanni Cappa1, Marzia Delorenzo1, Lorenzo Demitrya, Flavia Resta1, Gianmarco Secco1, Caterina Zattera1, Raffaele Bruno2, Stefano Perlini1 and Francesco Salinaro1*
1Emergency Medicine Unit and Emergency Medicine Postgraduate Training Program, University of Pavia, Pavia, ITALY
2Infectious Disease Unit, IRCCS Policlinico San Matteo Foundation, University of Pavia, Pavia, ITALY On behalf of the GERICO (Gruppo Esteso RIcerca COronarovirus) Lung Ultrasound Pavia Study Group: Ilaria Martino, Barbarah
Guglielmana, Ilaria Zunino, Federica Quaglia, Pietro Pettenazza, Serena Pioli di Marco, Anna Giulia Falchi, Claudia Alfano, Elisa Mossolani, Massimiliano Sciarrini, Igor Maisak, Michele Tassi, Stefano Galati, Vincenzo Capozza, Ilaria Melara, Benedetta Chiodi, Damiano Vignaroli, Lorenzo Scattaglia, Giulia Bissichini, Marco Bazzini.
*Corresponding author
Francesco Salinaro, IRCCS
Policlinico San Matteo Foundation,
University of Pavia
P.le Golgi, 19 27100 Pavia – Italy;
Tel: +39-0382-502470;
Fax: +39-0382-502400;