- 4192 Devonwood Way, Ashburn, Virginia, 20148
- Helpline: +1 (703) 665-3747
Amantadine, Parkinson’s Disease and COVID-19
- Home
- Back to Journal
- Article Details
Abstract
The lack of an effective vaccine against infection by acute respiratory syndrome coronavirus-2 [SARS-CoV-2] has resulted in renewed efforts aimed at the discovery of novel antiviral agents and, in many cases, the repurposing of existing compounds with established efficacy against other viruses for their efficacy against COVID-19. Amantadine started life as an effective antiviral therapy against influenza A by way of its actions targeting the viral M2 protein leading to altered replication. Amantadine was subsequently shown to be active against the SARS-CoV coronavirus by binding to a domain of protein E of that virus. COVID-19 is a novel coronaviral disease caused by SARS-CoV-2. Entry of the virus into the host cell requires binding of its “spike protein” to cellular receptors followed by its cleavage by host cell proteases such as Cathepsin L [CTSL]. A high throughput drug screen expression analysis of 466 drugs with potential to down-regulate CTSL gene expression was undertaken. Amantadine ranked 5th in potency leading to the hypothesis that interference with spike protein processing by amantadine could provide a useful therapy for COVID-19. The ED50 for amantadine was within the drug’s clinical pharmacokinetic profile. A serendipitous observation led to a pathophysiologic link between amantadine and Parkinson’s Disease [PD]. Subsequently, antibody responses to a range of coronaviruses were reported in CSF of PD patients and results of an early study showed benefit of amantadine for treatment of COVID-19 in 5 patients with PD. Two independent mechanisms are proposed as candidates with the potential for amantadine to mitigate the effects of COVID-19 namely 1] the down-regulation of host cell proteases leading to impaired genome release into the cytoplasm of the host cell and 2] its NMDA antagonist action leading to prevention of acute lung injury and respiratory distress.
Keywords: Amantadine, COVID-19, Parkinson’s Disease, SARS-CoV-2, spike protein, ACE-2 receptor, Cathepsin L, brain, CSF, acute respiratory failure, NMDA receptor
Introduction
The COVID-19 pandemic continues to spread throughout the world with the total number of confirmed cases passing the 6 million mark and attendant numbers of fatalities in excess of 375,000 at the time of writing of this article, a mortality rate somewhat greater than the 3.4% estimated by The World Health Organization [WHO].
In response to the pandemic, large numbers of Laboratories and Clinical Research Institutes in various parts of the world have become actively involved in the search for a vaccine. In view of the time required for the discovery, safety and efficacy testing and large-scale production of an eventual vaccine, other teams of investigators are concentrating their efforts on the discovery of novel antiviral agents and, in some cases, on the repurposing of existing agents with the potential to mitigate the symptoms of COVID-19.
A recent article reviews the currently-available drugs in the pipeline that are undergoing evaluation as possible candidates for treatment of COVID-19. The list is impressive and includes commercially-available antiviral agents, anti-malarial drugs, anti-inflammatory agents as well as traditional Indian medicines [1]. Some of the agents listed are currently undergoing the rigours of controlled clinical trials.
The present article focuses on the rationale and evidence in favour of the repurposing of amantadine for the prevention and treatment of COVID-19 by way of a review of the evidence derived from both experimental and clinical studies.
Chemical Structure and Established Antiviral Actions of Amantadine
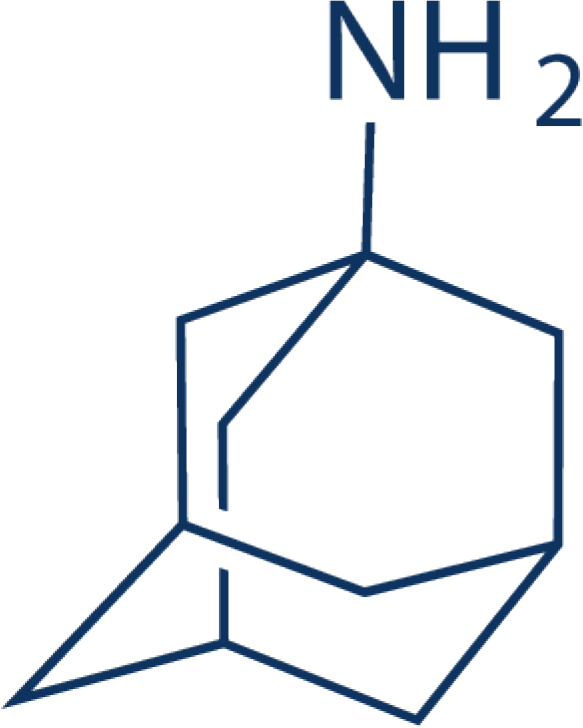
From a molecular structure standpoint, amantadine has an adamantane backbone with an amino group substituted at one of the four methyne positions resulting in the molecule 1-aminotricyclo [3.3.1.1.3.7] decane shown in figure 1.
Figure 1: Molecular space-filling structure of amantadine comprising an adamantine backbone with an amino group substituted at one of the four methyne positions giving rise to the molecule 1-aminotricyclo [3.3.1.1 3,7] decane [amantadine].
Amantadine has a relatively long association with the treatment of viral infections starting with a report of efficacy for the control of replication of the mouse hepatitis virus [2]. Importantly, since that time, amantadine has served as an effective antiviral therapy against influenza A by way of its actions targeting specifically the influenza A virus M2 protein having an essential role in the life cycle of the virus. Exposure to amantadine was shown to lead to blocking of the ion channel activity of the integral M2viral membrane protein with the capacity to affect viral replication [3].
In 2007, some 12 years prior to the discovery of COVID-19, a report appeared in which the novel SARS-CoV coronavirus responsible for the acute respiratory syndrome SARS was characterized [4]. It was subsequently demonstrated that this virus contains a small envelope protein known as protein E that is intimately involved in morphogenesis of the virus as well as in host cell apoptosis. It was demonstrated that amantadine binds to a pore formed by a lysine-flanked trans-membrane domain of protein E of the virus. Moreover, the action of amantadine occurred with relatively high affinity that was at least comparable to that reported for its effects on the M2 protein of the influenza A virus [5]. A more recent editorial suggested that, in view of its ability to inhibit viral replication by blockade of the influenza M2 ion channel required to deliver viral ribonucleoprotein into the host cytoplasm, amantadine would be an obvious target [6].
Amantadine and COVID-19
COVID-19 is caused by SARS-CoV-2 [Severe Acute Respiratory Syndrome Corona-virus-2] and virologists from The Chinese Academy of Sciences in Wuhan recently confirmed that the SARS-CoV-2 virus, like its predecessor, SARS-CoV attaches to the Angiotensin Converting Enzyme-2 [ACE-2] receptor. ACE-2’s functions include its action as a vasodilator that influences blood flow. The ACE-2 receptor is particularly common on the lung alveolar cells that line the pneumocytes [air sacs] in the lungs which is consistent with the common occurrence of respiratory symptoms and pneumonia associated with COVID-19 infections.
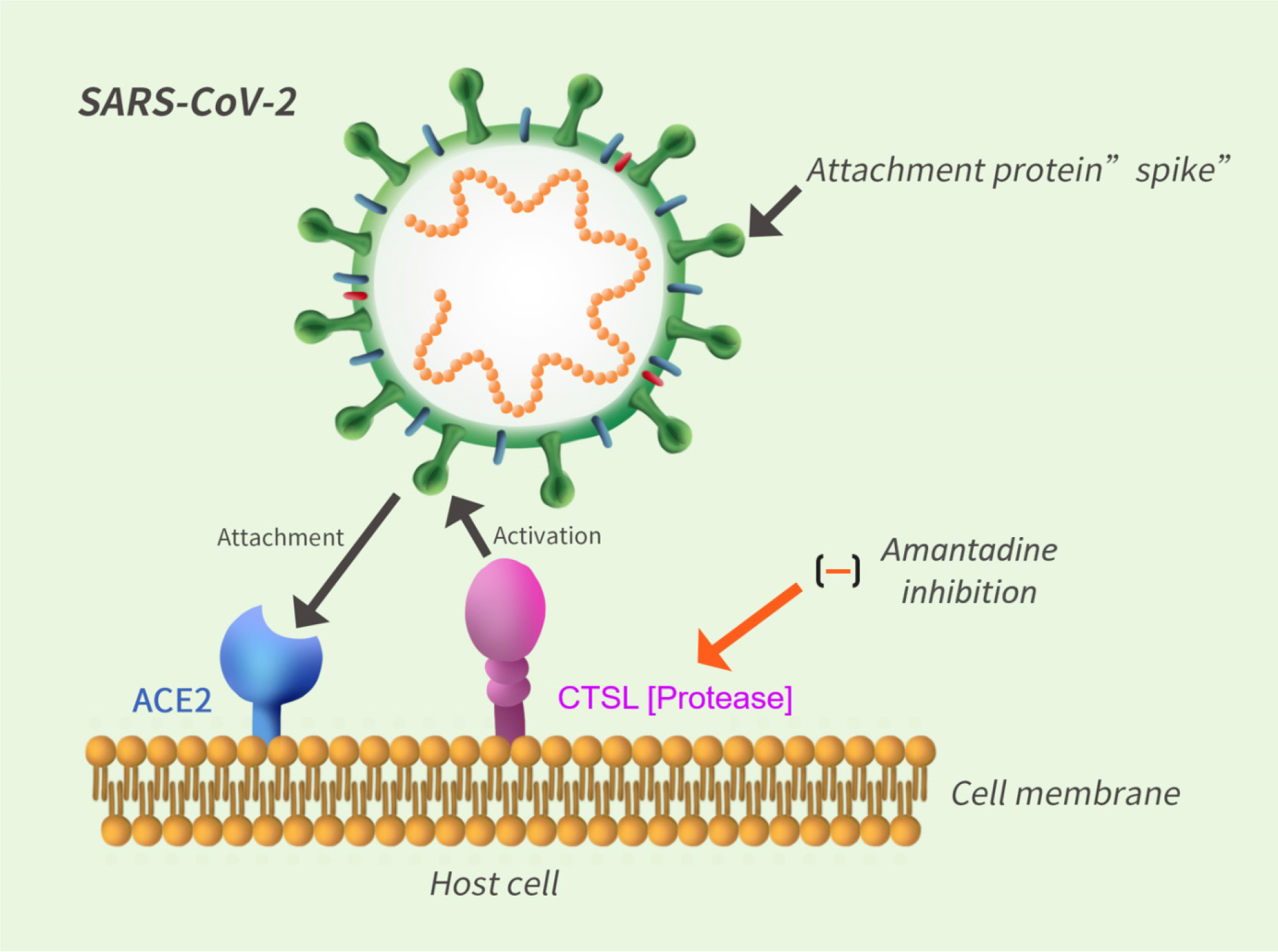
The entry of SARS-CoV-2 into the host cell takes place by binding of the viral “spike protein” to cellular receptors followed by cleavage of the spike protein by host cell proteases one example of which is CTSL. Each spike protein consists of 3 components that combine to form a trimer with 2 subunits, S-1 and S-2. Once attached to the ACE-2 receptor, the spike sheds its S-1 subunit and the remaining S-2 part changes its conformation in order to enable the viral envelope to fuse with the outer cellular membrane and to deposit the viral genome into the cytoplasm of the host cell [Figure 2].
Based upon previous information on the anti-viral action of amantadine, reports have started to appear suggesting the possibility that the drug may be of value for the mitigation of the effects of COVID-19 [7–8]. An appeal for the repurposing of agents of the adamantane family [including amantadine] for further evaluation as potentially useful agents for COVID-19 has recently been made [9].
Figure 2: Host cell protease dependence for SARS-CoV-2 infection. Fusion of viral and host cell proteins depends upon attachment of the viral spike protein to the ACE2 receptor on the host cell membrane followed by activation of a host cell protease such as Cathepsin L [CTSL] leading to fusion of viral and host cell membranes and release of viral genome into the host cell. Amantadine has the potential to down-regulate CTSL and decrease viral replication.
These reports were followed by an impressive study of the effects of a large number of compounds with the potential to down-regulate expression of the CTSL gene using a high throughput drug screen gene expression analysis and the retinal pigment epithelial cell line ARPE-19/HPV-16. One of the most potent agents with the capacity to down-regulate expression of the CTSL gene was amantadine [ranked 5th of 466 drugs tested]. This finding led the authors to conclude that interference with processing of the spike protein by the host cell either by alterations of the cellular environment or by modulation of lysosomal gene expression may offer a potentially useful therapeutic strategy for COVID-19 [10]. Moreover, the nature of the expression pattern of gene down-regulation by amantadine indicated a more generalized implication of lysosomes via a common set of transcription factors. It was proposed that these actions of amantadine, if effective, have the potential to result in reductions of viral load in SARS-CoV-2 positive patients with the likely consequences of decreased disease severity and progression. Of particular interest in the high throughput screen, it was noted that the effective dose of amantadine was within one order of magnitude of the agent’s clinical pharmacokinetic profile. Consequently, amantadine could be used within the current labelling guidelines [10]. Moreover, amantadine has the advantage of being an inexpensive, generic agent with side effects that are relatively mild compared to other putative COVID-19 treatments.
Amantadine, COVID-19 and Parkinson’s disease: What is the link?
Viruses have been implicated in causing encephalitis and Parkinson’s Disease including those responsible for the 1916–1930 epidemic [11] giving rises to what became known as “post-encephalitic Parkinson’s Disease”. The first report of an association between coronavirus and PD resulted from studies of antibody responses to four coronavirus antigens that included human coronaviruses 229E and OC43 in Cerebrospinal Fluid [CSF] of normal controls compared to patients with PD or with other neurological disorders. Patients with PD had significantly elevated CSF antibodies to coronaviruses compared to normal or other neurological disorders.
The link between viral infection, amantadine and PD began with the serendipitous observation recorded in 1968 when a 68-year old woman with moderately-severe PD reported that, while taking amantadine to prevent influenza symptoms noticed a remarkable remission of her tremor and cogwheel rigidity. These PD symptoms reappeared promptly upon cessation of amantadine. These observations resulted in a trial conducted in the following year in which 163 patients with PD were given amantadine; two thirds of patients showed significant clinical benefit [12].
Functional links between PD and COVID-19 come in many forms. Enhanced antibody responses to a range of coronaviruses have been reported in CSF of patients with PD [13] and basal ganglia lesions have been encountered in several other viral encephalides [14]. Moreover, results of an uncontrolled study suggest that amantadine may offer protection from COVID-19 in patients with a range of neurological disorders including five patients with PD [7].
PD is an age-related neurodegenerative disease characterized by progressive degeneration of dopaminergic neurons from the substantia nigra. Amantadine is beneficial by virtue of its actions in promotion of dopaminergic synaptic activity. Multiple mechanisms have been proposed including increases in dopamine synthesis, turnover and release. Studies using Positron Emission Tomography [PET] in humans with PD provide evidence consistent with the notion that increased synthesis of dopamine by amantadine occurs as a consequence of the antagonism of the NMDA subclass of glutamate receptors in brain [15]. Results of a systematic review with meta-analyses relating to the efficacy of amantadine for the treatment of PD and its associated dyskinesias are available [16].
Whether or not the NMDA receptor antagonist property of amantadine is implicated in the agent’s protective effects against COVID-19 or its neurological complications has not been assessed but there is evidence to support the notion that NMDA receptor antagonists may be beneficial in the treatment of other coronaviruses. For example, the human coronavirus strain HCoV-OC43 is known to infect and persist in human neural cells where it activates neuroinflammatory and neurodegenerative mechanisms and treatment with HCoV-OC43-infected mice with the non-competitive NMDA receptor antagonist memantine, an agent that is structurally-related to amantadine, has been found to limit viral replication and to improve neurological symptoms [17]. Moreover, the expression of NMDA receptors in the lungs and airways [18] may underpin the signalling mechanisms implicated in the pathogenesis of acute lung injury and the acute respiratory distress syndrome that is characteristic of COVID-19.
Cascade of CNS involvement in COVID-19
The pathophysiologic link between PD and COVID-19 is not the only example of the impact of the virus on CNS function. There is evidence to suggest that COVID-19 infections modify CNS function in a systematic and widespread manner. In a retrospective study of 214 hospitalized COVID-19 patients in Wuhan, China, 36.4% manifested neurological symptoms with more severely affected patients showing signs of impaired consciousness, acute cerebrovascular disease and skeletal muscle injuries [19]. Almost 90% of patients with severe COVID-19 manifest a loss of taste and smell in addition to hypoxic encephalopathy related to acute respiratory distress.
A cascade of CNS involvement in COVID-19 has been proposed whereby the dissemination of SARS-CoV-2 in the systemic circulation [or alternatively across the cribriform plate that is equipped with deep grooves supporting the olfactory bulb and perforated by olfactory foramina that allow the passage of the olfactory nerves] leads to involvement of the CNS [20].
ACE-2 receptors are expressed widely in the CNS including brainstem cardio-respiratory neurons [21] and based on the results of experimental and clinical studies, it was suggested that neuro-invasive effects of SARS-CoV-2 may play a role in the pathogenesis of the respiratory failure consistently encountered in COVID-19 patients [22].
Conclusion and Future Studies
Based upon the evidence presented it is reasonable to conclude that amantadine has the potential to mitigate the effects of COVID-19 and that it may do so by two independent mechanisms, namely:
Down-regulation of the expression of host cell proteases such as [but likely not necessarily limited to] CTSL resulting in impairment of the fusion of viral and host cell membranes leading to impaired release of the viral genome into the cytoplasm of the host cell. Decreased viral entry has the potential to decrease viral load in SARS-CoV-2 infected patients with consequent improvement in clinical outcomes. Appropriately-designed, adequately-powered controlled clinical trials are now required in order to assess the therapeutic efficacy of amantadine for the treatment of COVID-19 infection.
Amantadine is a potent non-competitive NMDA receptor antagonist. Experimental studies with coronaviruses reveal that such agents have the capacity to limit viral replication and improve neurological outcomes. NMDA receptors are localized in multiple sites in mammalian lungs and airways and a role for these receptors has been proposed for the activation of NMDA receptors in some forms of acute lung injury [23] as well as in the pathogenesis of the acute respiratory distress syndrome [18]. Moreover, it has been suggested that the neuro-invasive potential of SARS-CoV-2 may play a role in the pathogenesis of the acute respiratory failure in COVID-19 patients [22]. Further studies [both preclinical and clinical] are now required in order to examine these possibilities.
Funding
Expenses for laboratory and clinical studies and for publication costs incurred by the author were provided by The Canadian Institutes of Health Research [CIHR] and The University of Montreal [CHUM].
References
- Vellingiri B, Jayaramayya K, Iyer M, Arul N, Vivekanandhan G, et al. (2020) COVID-19: A promising cure for the global panic. Sci Total Environ 725: 138277. [View]
- Leibowitz JL, Reneker SJ (1993) The effect of amantadine on mouse hepatitis virus replication. Adv Exp Med Biol 342: 117–122. [View]
- Wang C, Takeuchi K, Pinto LH, Lamb RA (1993) Ion channel activity of influenza a virus M2 protein: characterization of the amantadine block. J Virol 67: 5585–5594. [View]
- Rota PA, Oberste MS, Monroe SS, Nix WA, Campagnoli R, et al. (2003) Characterization of a novel coronavirus associated with severe acute respiratory syndrome. Science 300: 1394–1399. [View]
- Torres J, Maheswari U, Parthasarathy K, Ng L, Liu DX, et al. (2007) Conductance and amantadine binding of a pore formed by a lysine-flanked transmembrane domain of SARS coronavirus envelope protein. Protein Sci 16: 2065–2071. [View]
- Stoessl AJ, Bhatia KP, Merello M (2020) Movement Disorders in the World of COVID-19. Mov Disord Clin Pract 7: 355–356. [View]
- Rejdak K, Grieb P (2020) Adamantanes might be protective from COVID-19 in patients with neurological diseases: multiple sclerosis, parkinsonism and cognitive impairment. Mult Scler Relat Disord 42: 102163. [View]
- Abreu GEA, Aguilar MEH, Covarrubias DH, Durán FR (2020) Amantadine as a drug to mitigate the effects of COVID-19. Med Hypotheses 40: 109755. [View]
- Cimolai N (2020) Potentially repurposing adamantanes for COVID-19. J Med Virol 2020 [Online ahead of print]; 10.1002/jmv.25752. [View]
- Smieszek SP, Przychodzen BP, Polymeropoulos MH (2020) Amantadine disrupts lysosomal gene expression: A hypothesis for COVID19 treatment [published online ahead of print, 2020 Apr 30]. Int J Antimicrob Agents. 2020; 106004. [View]
- Duvoisin RC, Yahr MD (1965) Encephalitis and Parkinsonism. Arch Neurol 12: 227–239. [View]
- Schwab RS, England AC Jr, Poskanzer DC, Young RR (1969) Amantadine in the treatment of Parkinson’s disease. JAMA 208: 1168–1170. [View]
- Fazzini E, Fleming J, Fahn S (1992) Cerebrospinal fluid antibodies to coronavirus in patients with Parkinson’s disease. Mov Disord 7: 153–158. [View]
- Fishman PS, Gass JS, Swoveland PT, Lavi E, Highkin MK, et al. (1985) Infection of the basal ganglia by a murine coronavirus. Science 229: 877–879. [View]
- Deep P, Dagher A, Sadikot A, Gjedde A, Cumming P (1999) Stimulation of dopa decarboxylase activity in striatum of healthy human brain secondary to NMDA receptor antagonism with a low dose of amantadine. Synapse 34: 313–318. [View]
- Kong M, Ba M, Ren C, Yu L, Dong S et al. (2017) An updated meta-analysis of amantadine for treating dyskinesia in Parkinson’s disease. Oncotarget 8: 57316–57326. [View]
- Brison E, Jacomy H, Desforges M, Talbot PJ (2014) Novel treatment with neuroprotective and antiviral properties against a neuroinvasive human respiratory virus. J Virol 88: 1548–1563. [View]
- Dickman KG, Youssef JG, Mathew SM, Said SI (2004) Ionotropic glutamate receptors in lungs and airways: molecular basis for glutamate toxicity. Am J Respir Cell Mol Biol 30: 139–144. [View]
- Mao L, Jin H, Wang M, et al. (2020) Neurologic Manifestations of Hospitalized Patients With Coronavirus Disease 2019 in Wuhan, China [published online ahead of print, 2020 Apr 10]. JAMA Neurol e201127. [View]
- Baig, Khaleeq A, Ali U, Syeda H (2020) Evidence of the COVID-19 Virus Targeting the CNS: Tissue Distribution, Host-Virus Interaction, and Proposed Neurotropic Mechanisms. ACS Chem Neurosci 11: 995–998. [View]
- Doobay MF, Talman LS, Obr TD, Tian X, Davisson RL, et al. (2007) Differential expression of neuronal ACE2 in transgenic mice with overexpression of the brain renin-angiotensin system. Am J Physiol Regul Integr Comp Physiol. 292: R373–R381. [View]
- Li YC, Bai WZ, Hashikawa T (2020) The neuroinvasive potential of SARS-CoV2 may play a role in the respiratory failure of COVID-19 patients. J Med Virol 2020. [View]
- Said SI (2005) Glutamate Receptor Activation in the Pathogenesis of Acute Lung Injury. In: Bhattacharya J. (eds) Cell Signaling in Vascular Inflammation. Humana Press. [View]
Article Type
Review Article
Publication history
Received: June 02, 2020
Accepted: June 09, 2020
Published: June 11, 2020
R-Infotext Citation:
Roger F Butterworth (2020) Amantadine, Parkinson’s disease and COVID-19. Clar J Infect Dis Ther 01(01): 7–12.
Roger F Butterworth*
Department of Medicine, University of Montreal, Canada
*Corresponding author
Roger F Butterworth PhD DSc,
Professor of Medicine,
Department of Medicine,
University of Montreal,
45143 Cabot Trail,
Englishtown, NS, B0C 1H0,
Canada;