- 4192 Devonwood Way, Ashburn, Virginia, 20148
- Helpline: +1 (703) 665-3747
A Comprehensive COVID-19 Meta-Analysis: Clinical Data of 18,450 Patients
- Home
- Back to Journal
- Article Details
Abstract
Purpose: It is very important to determine the clinical features of COVID-19 and to present and collect global data of these features. In this meta-analysis, it was aimed to examine the published studies and to collect and analyze the data of COVID-19 patients.
Method: We scanned in PubMed and Google Scholar and the studies involving more than 40 patients and examining the clinical, laboratory, and radiology findings of COVID-19 patients and included in the meta-analysis. Other meta-analyses are also examined in this meta-analysis.
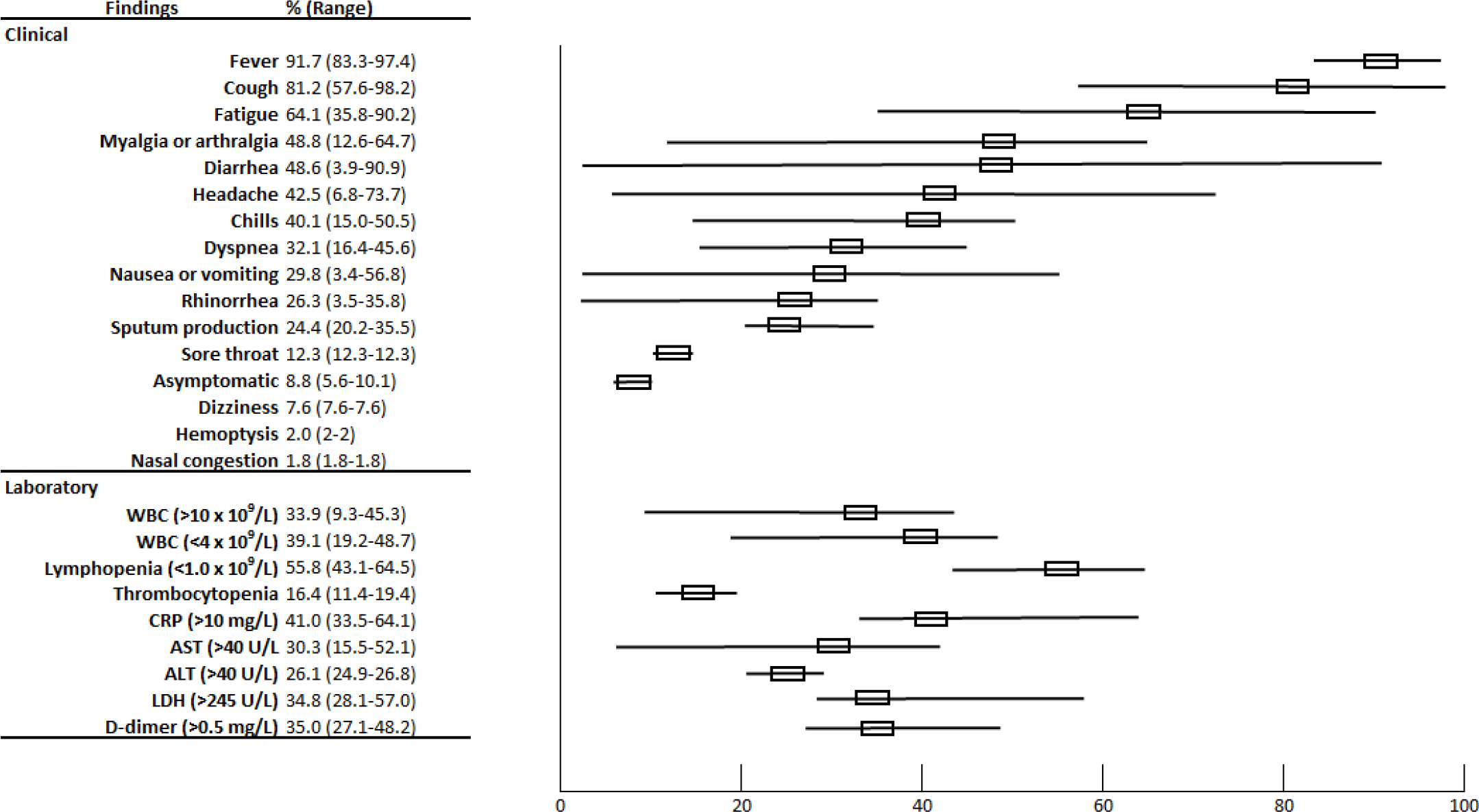
Findings: By the end of this meta-analysis we covered 61 articles covering a total of 18, 450 patients. According to the analyses, it is detected that the most common findings of COVID- 19 patients were fever (91.7%), dry cough (81.2%), fatigue (64.1%), myalgia or arthralgia (48.8%) and diarrhea (48.6%). The high fever rate was determined as 23.2%. It is observed that the most common laboratory findings in COVID-19 cases were lymphopenia (55.8%), high C-reactive protein (CRP) levels (41.0%), and leukopenia (39.1%). The most common radiological finding was detected to be ground-glass opacity (32.4%). The bilateral lung involvement rate was determined as 16.2%.
Result: In this meta-analysis, it is shown that the symptoms such as fever, cough, and fatigue were the most common in COVID-19 patients, laboratory findings were supportive but radiological findings were diagnostic, and the diagnosis was confirmed by Real-Time PCR.
Keywords: COVID-19, SARS-CoV-2, Clinical features, Ground-glass Opacity, Adult
Introduction
Severe Acute Respiratory Syndrome (SARS) coronavirus-2 (SARS-CoV-2), an RNA virus called new coronavirus, has fast and high infectious properties. Coronavirus-2019 disease (COVID-19) caused by SARS-CoV-2 caused a huge pandemic that is spreading rapidly throughout the world. The COVID-19 epidemic, which started in Wuhan, China in December 2019, spread in China in a short period of time, then spread to other countries and reached a picture that caused millions of cases and hundreds of thousands of deaths in about 6 months [1–3].
COVID-19 causes respiratory infection. However, COVID-19 can manifest itself differently than typical respiratory infections with various combinations of clinical signs and symptoms. In some cases, it is quite difficult to tell COVID-19 apart from a viral upper respiratory tract infection. In most cases, the course of the disease is asymptomatic and sometimes it may occur with a symptom that is different than of a typical respiratory infection [1–5]. For these reasons, it is of great importance to determine the clinical features of COVID-19 and to collect and present the global data of these features. In this meta-analysis, it was aimed to examine the published studies and to collect and analyze the data of COVID-19 patients.
Material and Method
This meta-analysis was planned and conducted in accordance with the Preferred Reporting Items for Meta-Analysis of Observational Studies in Epidemiology Statement (PRISMA) criteria [6].
Research Strategy
The studies to be included in the meta-analysis were scanned in PubMed and Google Scholar. While scanning, the terms ‘Coronavirus’, ‘Coronavirus’, ‘2019-nCoV’, ‘COVID-19’ or ‘SARS- CoV-2’ were used.
Criteria for the Inclusion to Meta-Analysis
We only included published studies or the studies accepted for publication in this meta- analysis. Screening results before 2020 were eliminated. Case reports are not included in the meta-analysis, as they will not contribute substantially to the meta-analysis. Studies involving patients with less than 40 patients were not included in the meta-analysis, as this may impair the standardization of the data. No language restriction was implemented. We only included studies investigating clinical, laboratory, and radiology findings of COVID-19 patients in the meta-analysis.
Article Selection and Data Collection
For the first step, the titles and summaries of the selected articles were examined in terms of suitability for the meta-analysis. In the next stage, we reviewed the full text of the selected articles. Articles outside the focus subject of the meta-analysis were eliminated. Other meta- analyses [7–12] were included in this meta-analysis with the view that they would contribute significant numbers and statistics, but duplicated articles and data that were examined in meta-analyses in advance were excluded. As a result, the data of the selected articles were collected.
Statistical analysis
All statistical analyses were performed using SPSS 25.0 software (IBM SPSS, Chicago, IL, USA) and online calculators were appropriate. Descriptive data is given as numbers and percentages. Original incidence rates (r) were transformed to rate tr, and were used in single- arm meta-analysis. The heterogeneity between studies was analyzed using a c2 test (p<0.10) and quantified using the I2 statistic.
Findings
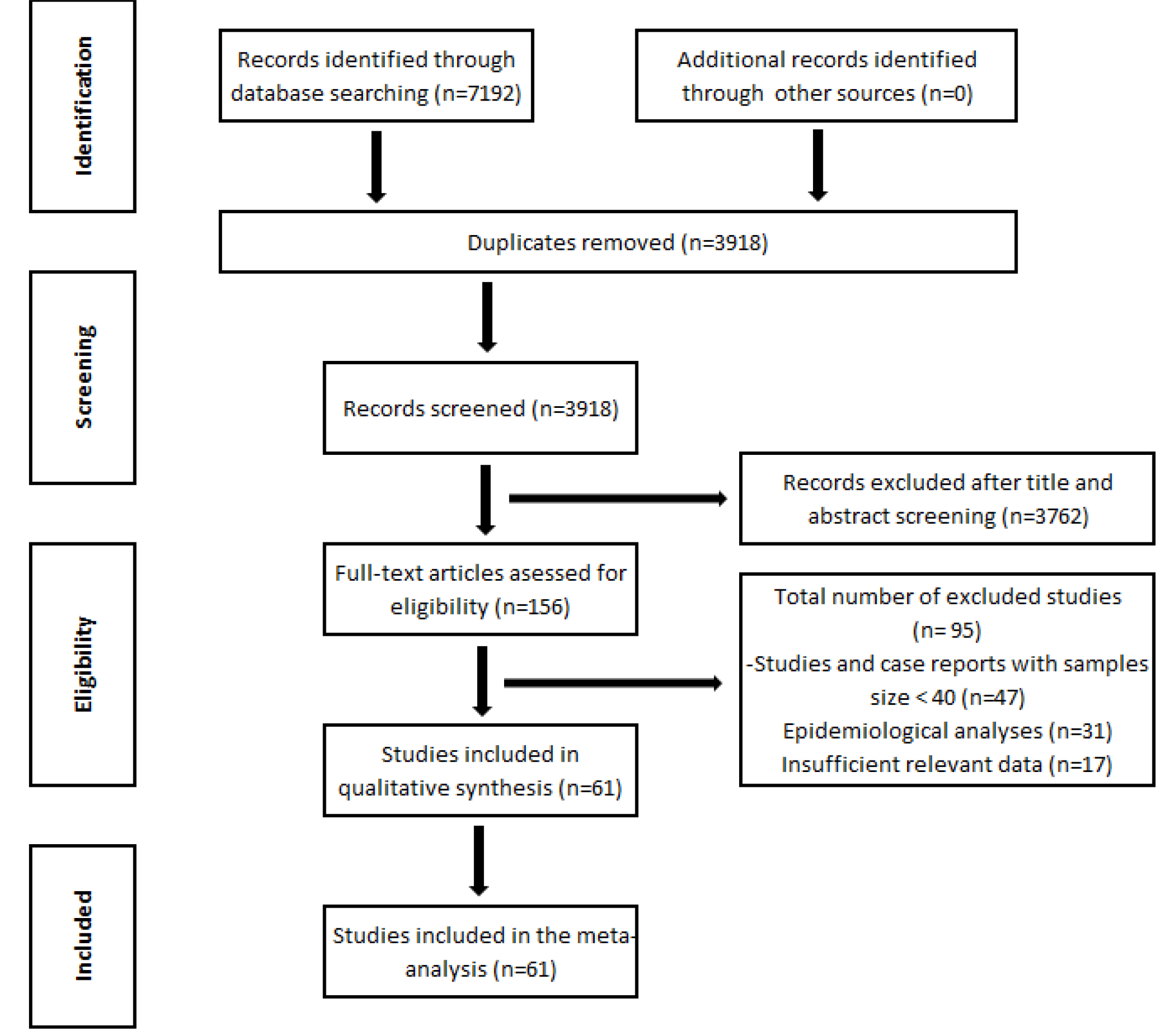
By the end of the scan, a total of 7192 articles were found. The duplicated ones were excluded. Then, the summaries of the channel studies were examined and those that were not related to our subject or those who could not contribute to our study were eliminated. The full texts of the remaining articles were examined and 61 articles [13–73] containing a total of 18, 450 patients matching our criteria was included in the meta-analysis (Graph 1).
Graph 1: Flow algorithm of the literature review process.
According to the analyses, it is detected that the most common findings of COVID-19 patients were fever [91.7%], dry cough (81.2%), fatigue (64.1%), myalgia or arthralgia [48.8%] and diarrhea (48.6%) (Table 1). The high fever rate was determined as 23.2%.
Table 1: Rates of clinical signs and laboratory findings in COVID-19 patients.
|
Findings |
%(Range) |
|
Clinical |
|
|
Fever |
91.7 (83.3–97.4) |
|
Cough |
81.2 (57.6–98.2) |
|
Fatigue |
64.1 (35.8–90.2) |
|
Myalgia or arthralgia |
48.8 (12.6–64.7) |
|
Diarrhea |
48.6 (3.9–90.9) |
|
Headache |
42.5 (6.8–73.7) |
|
Chills |
40.1 (15.0–50.5) |
|
Dyspnea |
32.1 (16.4–45.6) |
|
Nausea or vomiting |
29.8 (3.4–56.8) |
|
Rhinorrhea |
26.3 (3.5–35.8) |
|
Sputum production |
24.4 (20.2–35.5) |
|
Sore throat |
12.3 (12.3–12.3) |
|
Asymptomatic |
8.8 (5.6–10.1) |
|
Dizziness |
7.6 (7.6–7.6) |
|
Hemoptysis |
2.0 (2–2) |
|
Nasal congestion |
1.8 (1.8–1.8) |
|
Laboratory |
|
|
WBC (>10 x 109/L) |
33.9 (9.3–45.3) |
|
WBC (<4 x 109/L) |
39.1 (19.2–48.7) |
|
Lymphopenia (<1.0 x 109/L) |
55.8 (43.1–64.5) |
|
Thrombocytopenia |
16.4 (11.4–19.4) |
|
CRP (>10 mg/L) |
41.0 (33.5–64.1) |
|
AST (>40 U/L) |
30.3 (15.5–52.1) |
|
ALT (>40 U/L) |
26.1 (24.9–26.8) |
|
LDH (>245 U/L) |
34.8 (28.1–57.0) |
|
D-dimer (>0.5 mg/L) |
35.0 (27.1–48.2) |
It is observed that the most common laboratory findings in COVID-19 cases were lymphopenia (55.8%), high C - reactive protein (CRP) levels (41.0%), and leukopenia (39.1%). (Table 1) (Graph 2).
Graph 2: Rates clinical symptoms and laboratory findings in COVID-19 patients.
The most common radiological finding was detected to be ground-glass opacity [32.4%]. The bilateral lung involvement rate was determined as 16.2%.
Discussion
The COVID-19 pandemic has spread rapidly throughout the world. This spread occurred in just a few months. Therefore, clinical information on COVID-19 has not yet reached a sufficient level. There are still not enough studies, reports, and evidence about the clinical, laboratory, and radiology findings of COVID-19. Accordingly, it is not easy to distinguish COVID-19 from other respiratory diseases at the time of admission [1–5].
COVID-19 started in China first and spread rapidly throughout the country. After the COVID- 19 case number reached its peak in China, it spread to other countries, and then a pandemic developed. The number of COVID-19 reports of researchers, whose first experience was in China, increased rapidly [1–5]. Accordingly, most of the COVID-19 studies in the literature originate from China. Large-scale case reports from Europe and the USA are not yet sufficient. In some studies, it was reported that the clinical picture in the first months of the COVID-19 pandemic varied in the following months, the variety of symptoms has differed, and there was a difference in the rates of severe disease [3–5, 7–10]. Correspondingly, clinical analyses, whose most initial data is from China, may not be sufficient to clearly show the clinical situation in other parts of the world. Comprehensive and wide-ranging studies and analyses can prevent this negative situation. Our study covers data from a total of 18, 450 COVID-19 patients, and as far as we know [https://www.ncbi.nlm.nih.gov/; access date: 16/06/2020], our study is the most comprehensive meta-analysis ever conducted.
Clinic
Fever
In this meta-analysis, fever was found to be the most common symptom in COVID-19 patients. Other meta-analyses reported the rate of fever in the range of 83.3% and 97.4% [7–12]. In our study, the rate of fever was found to be 91.7%. This finding means that fever is common enough to be diagnostic for patients with COVID-19. Accordingly, COVID-19 should be among the first diagnoses for a patient with suspected inflammatory respiratory tract infection.
The level of fever is as important as its occurrence in COVID-19 patients. The high fever rate in COVID-19 patients is much lower than bacterial respiratory infections. Some viral respiratory infections also often cause higher fever. Some studies have accepted the high fever threshold as 37.5 ° C, and others as 38 ° C. The high fever rate is not very elevated in COVID-19 cases according to both threshold values [1–5, 10–14]. In this meta-analysis, the high fever rate was determined as 23.2%. These findings indicate that mild fever is seen more frequently in COVID-19, and high fever should not be expected especially in new patients.
Cough
Cough is another most common symptom in patients with COVID-19. It was reported that dry cough was observed in the early period of the COVID-19 clinical picture, and people with advanced disease developed a productive cough in the following weeks [2–4, 12–15]. In this meta-analysis, dry cough was the second most common symptom in COVID-19 cases. In other meta-analyses, dry cough rates were reported to be between 57.6% and 98.2% [7–12]. In this meta-analysis, the rate of dry cough is found to be 81.2 %. According to these findings, the dry cough should be expected in patients with suspected COVID-19. COVID-19 should be among the first diagnoses for patients with suspected upper respiratory tract infection with dry cough.
In patients with COVID-19, sputum production occurs after the first week depending on if the infection reaches the lower respiratory tract and lungs. Productive cough develops in these patients during this period [3–5]. The productive cough rate has been reported to be between 20.2% and 35.5% in other meta-analyses [7–12]. In this meta-analysis, the productive cough rate was determined as 24.4%. According to this data, it should be kept in mind that COVID-19 patients may develop significantly in the following weeks of the disease, although no signs of productive cough are expected at the first admission. COVID-19 should also be kept in mind in patients admitted with a productive cough.
Fatigue
It has been stated that the complaint of fatigue is high in COVID-19 cases. The question of whether the patient feels fatigue may not be as common as if they have more concrete symptoms such as fever and cough. Accordingly, the presence of fatigue, a more subjective symptom, may be ignored by both the patient and the clinician in some cases. In fact, fatigue in young children is a symptom that cannot be expressed by the patient [1–5, 7–9]. Therefore, the rates of fatigue reported in COVID-19 cases may have been calculated lower than they should have been. This situation should be taken into account. In other meta-analyses, the rate of COVID-19 cases with fatigue complaint was in the range of 35.8% and 90.2% [7–12]. Given this situation, this range is very wide and may be due to the above-mentioned reasons. In this meta-analysis, the rate of fatigue was determined to be 64.1% in COVID-19 patients. According to these findings, fatigue should be expected in COVID-19 patients. However, considering that the complaint of fatigue is a common condition in most diseases with or without infection, it may be thought that fatigue is not a specific symptom for COVID-19.
Dyspnea
It was stated that shortness of breath is not seen frequently in COVID-19 cases, mostly seen in progressive disease states, but this symptom is highly specific for COVID-19. Shortness of breath is not a highly anticipated symptom of COVID-19 patients during the first admission to the hospital. However, in cases where the disease progresses and reaches the lower respiratory tract and lungs, shortness of breath and respiratory failure may develop due to alveolar damage [1–5]. In other meta-analyses, the rate of shortness of breath in COVID-19 cases was reported in the range of 16.4% and 45.6% [7–12]. The wide range of this may be due to the fact that some studies cover only advanced COVID-19 cases, and some studies were conducted in regions where the pandemic started and was most severe. In this meta-analysis, the rate of shortness of breath was determined as 32.1%. These findings show that in patients with shortness of breath, COVID-19 should be included in the diagnoses that come to mind, but in patients with COVID-19, shortness of breath, in general, is not expected at the first admission.
Other upper respiratory tract infection symptoms
A typical viral upper respiratory tract infection picture is seen in most of the COVID-19 cases. These patients may experience nasal discharge or congestion, sore throat, and headache in various combinations. These symptoms are usually moderate and not very determinant for diagnosis [1–5]. In other meta-analyses, the frequency of headaches has been reported to be between 6.8% and 72.3%, and nasal discharge between 3.5% and 35.8% [7–12]. This wide range shows the variability of these symptoms. In this meta-analysis, the rate of nasal discharge was determined as 26.3%, headache rate as 42.5%, and sore throat as 12.5%. These findings show that the rate of typical upper respiratory symptoms can vary greatly in COVID- 19 patients, but the specificity of these symptoms for COVID-19 is very low.
Gastrointestinal System Symptoms
In some COVID-19 cases, symptoms of the Gastrointestinal System (GIS) such as nausea, vomiting, and diarrhea can be seen. In some cases, it has been reported that GIS symptoms can be seen without any symptoms of respiratory infection [1–5]. In other meta-analyses, nausea and/or vomiting rate has been reported in a wide range between 3.4% and 56.8% [7–12]. In other meta-analyses, the rate of diarrhea was reported in a range covering almost every possibility, from 3.9% to 90.0% [7–12]. In this meta-analysis, the rate of nausea-vomiting was determined as 29.8%, and diarrhea rate as 48.6%. These findings show that GIS symptom rates can differ greatly in COVID-19 cases, and the sensitivity and specificity values of these symptoms for COVID-19 may be very low. However, these findings also point out the importance of avoiding the COVID-19 diagnosis for the patients admitted to the hospital with GIS symptoms.
Laboratory
In the COVID-19 table, it has been reported that the laboratory findings also vary greatly. None of the laboratory findings have high specificity for the COVID-19 diagnosis. However, laboratory findings can still provide useful data when evaluated together with clinical and radiological findings [1–5].
Leukocyte Count
In most COVID-19 cases, leukocyte counts can be affected. While leukocytosis is seen in some cases, some may develop leukopenia. These different tables can also vary according to the clinical condition of the patients, the severity and stage of the disease [1–5]. In other meta- analyses, the leukocytosis rate has been reported in a wide range of 9.3% and 45.3% [7–12]. The leukopenia rate has been reported in a wide range between 19.2% and 48.7%. In this meta-analysis, the leucocytosis rate was determined as 33.9%, and the leukopenia rate as 39.1%. All these findings indicate that abnormal leukocyte levels can be expected in COVID- 19 cases, but the leukocyte level is not determinant for the COVID-19 diagnosis. Leukocyte levels may still be helpful in patients admitted to the hospital with suspected COVID-19.
Lymphocyte levels
Lymphopenia has been reported in COVID-19 cases at substantial rates. Lymphocytes have been reported to be adversely affected by infection and tend to fall. Since viral infections are generally expected to increase in lymphocyte levels, lymphopenia may be supportive for the COVID-19 diagnosis [1–5]. The lymphopenia rate has been reported in the range of 43.1% and 64.5% in other meta-analyses [7–12]. In this meta-analysis, the lymphopenia rate was determined at a significant rate of 55.8%. All these data show that the development of lymphopenia can be expected in patients with COVID-19 and COVID-19 should be among the infections that should be considered first in patients with lymphopenia.
C-reactive protein (CRP) level
It has been reported that an increase in C-reactive protein (CRP) level can be observed in COVID-19 cases due to inflammatory reactions [1–5]. In other meta-analyses, the high CRP level rate has been reported in the range of 33.5% and 64.1% [7–12]. In this meta-analysis, the high CRP level was determined as 41.0% which is a significant number. These findings mean that the detection of a high CRP level in COVID-19 patients supports the diagnosis. However, high CRP levels are not specific for COVID-19 since it is known that the CRP level increases in all inflammatory conditions.
Liver Enzymes
In COVID-19 cases, liver damage may also occur, especially when the disease progresses and the level of liver enzymes such as aspartate aminotransferase (AST) and alanine aminotransferase (ALT) may elevate [1–5]. In other meta-analyses, high AST levels have been reported in a wide range of 15.5% and 52.1% and high ALT levels in the range of 24.9% and 26.8% [7–12]. In this meta-analysis, AST levels were determined as 30.3%, and ALT levels as 26.1%. These findings show that the increase in liver enzymes should not be expected in patients with COVID-19 especially at the first admission to the hospital. However, according to these findings, COVID-19 should be among the diseases that should come to mind in respiratory tract infection patients with high ALT and/or AST levels.
Serum lactate dehydrogenase levels
It has been reported that the level of lactate dehydrogenase enzyme may also increase in COVID-19 cases [1–3]. In meta-analyses, the LDH increase rate was reported in the range of 28.1% and 57.0% [7–12]. In this meta-analysis, the LDH increase rate was determined as 34.8%. These findings mean that LDH increase may support the COVID-19 diagnosis.
Radiological Findings
Radiological imaging plays an important role for the COVID-19 diagnosis. Radiological examination results provide very important data in the diagnosis of COVID-19. It was reported that the rate of Ground-glass Opacity detected in high rates in the imaging results of asymptomatic patients, especially without any respiratory symptoms [1–5].
Ground-Glass Opacity
Ground-glass opacity is observed in COVID-19 cases, especially in cases where the infection has reached the lower respiratory tract and the lungs and caused a pathological condition due to alveolar damage and subsequent inflammatory reactions. Ground glass opacity is the most common finding provided by lung imaging in COVID-19 cases [1–5]. In other meta-analyses, ground-glass opacity has been reported in the range of 18.5% and 43.6% [7–12]. In this meta- analysis, the ground-glass Opacity rate was determined as 32.4%. These findings show that ground-glass opacity is very important in the COVID-19 diagnosis and its specificity is high. These findings also indicate that ground-glass opacity should be expected in most of the advanced cases and in cases where ground-glass opacity is detected, the patient must be tested for COVID-19.
Bilateral lung involvement
Lung involvement is frequently seen in COVID-19 cases. The rate of lung involvement increases, especially in advanced cases. It has been reported that a significant part of lung involvement is bilateral [1–5]. In other meta-analyses, the rate of detection of bilateral lung involvement detected through radiological imaging was reported in the range of 12.4% and 24.1%. In this meta-analysis, the bilateral lung involvement rate was determined as 16.2%. These findings show that COVID-19 patients should be monitored closely for lung involvement.
COVID-19 Diagnosis
The diagnosis of COVID-19 is made after the clinical, laboratory, and radiology findings are evaluated together. Having a history of close contact with someone diagnosed with COVID- 19 or someone with a clinical picture similar to the COVID-19 chart is among the most important indications for conducting COVID-19 tests. The people who have a history including any contact with a COVID-19 patient should be tested even when they are asymptomatic, in terms of both detecting the infection and preventing possible contamination under quarantine measures. Apart from this, living in an area, or staying somewhere where the COVID-19 outbreak is common is a situation that reinforces the suspicion of COVID-19. Besides these stories, the presence of 1 or 2 of the above-mentioned clinical findings raises suspicion for COVID-19. In addition to clinical findings, in case of abnormal laboratory findings or abnormal radiological findings, testing are recommended for COVID-19 [74–78].
In order to confirm the diagnosis of COVID-19, lower respiratory tract samples such as nasopharyngeal and oropharyngeal swabs or flush samples should be collected; as well as lower respiratory samples such as endotracheal aspirate or bronchoalveolar lavage and/or sputum especially for respiratory patients. It has been reported that the nasopharyngeal swab sample can give more accurate results as the density of the virus can be high in the nasal cavity mucosa [74–78].
The sample should be delivered to the laboratory quickly and under suitable conditions, the laboratory should be contacted prior to the sample transfer process, and the sample should not be kept in the laboratory. The sample can be frozen at -20 ° C or -70 ° C in case of possible delays or additional serological tests to be made in the future [74–78].
Confirmation of the diagnosis is made by detecting SARS-CoV-2. The viral RNA of SARS- CoV-2 is detected by amplifying with real-time reverse transcriptase polymerized chain reaction [RT-PCR] method. In addition, the diagnosis can be confirmed by genome sequencing conducted to see the viral RNA genome. Additionally, the detection of virus- specific IgM and IgG antibodies through using serological methods is also diagnostic. The presence of antibodies without viral RNA indicates that the patient most likely already had the infection and recovered from it. Situations where viral RNA is detected but the antibody cannot be detected indicate that the patient has active COVID-19 and highly contagious. If viral RNA is negative, the test should be repeated. The negative re-test results do not rule out possible COVID-19 diagnosis completely. Pseudo-negativity in PCR tests may be due to different reasons such as clinical sample not being taken or processed appropriately or viral mutation. If the cases are still suspected with COVID-19, chest computerized tomography may be diagnostic [74–78].
Result
In this meta-analysis, it was demonstrated that the symptoms of fever, cough, and fatigue were the most common symptoms of COVID-19 patients, laboratory findings were supportive but radiological findings were diagnostic, and the diagnosis was confirmed by RT-PCR.
References
- Shereen MA, Khan S, Kazmi A, Bashir N, Siddique R (2020) COVID-19 infection: Origin, transmission, and characteristics of human coronaviruses. J Adv Res 24: 91?98. [View]
- Jin Y, Yang H, Ji W, Wu W, chen S, et al. (2020) Virology, Epidemiology, Pathogenesis, and Control of COVID-19. Viruses 12: 372. [View]
- Huang X, Wei F, Hu L, Wen L, Chen K (2020) Epidemiology and Clinical Characteristics of COVID-19. Arch Iran Med 23: 268–271. [View]
- Pascarella G, Strumia A, Piliego C, Bruno F, Del Buono R, et al. (2020) COVID-19 diagnosis and management: a comprehensive review [published online ahead of print, 2020 Apr 29]. J Intern Med 2020. [View]
- Singhal T (2020) A Review of Coronavirus Disease-2019 (COVID-19). Indian J Pediatr 87: 281–286. [View]
- Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gøtzsche PC, et al. (2009) The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. PLoS Med 6: e1000100. [View]
- Rodriguez-Morales AJ, Cardona-Ospina JA, Gutiérrez-Ocampo E, Villamizar-Peña R, Holguin-Rivera Y, et al. (2020) Latin American Network of Coronavirus Disease 0-COVID- Research (LANCOVID-). Electronic address: https://www.lancovid.org. Clinical, laboratory and imaging features of COVID-: A systematic review and meta-analysis. Travel Med Infect Dis 34: 101623. [View]
- Zhu J, Zhong Z, Ji P, Li H, Li B, Pang J, et al. (2020) Clinicopathological characteristics of patients with COVID- in China: a meta-analysis. Fam Med Community Health 8: e000406. [View]
- Zheng Z, Peng F, Xu B, Zhao J, Liu H, et al. (2020) Risk factors of critical & mortal COVID- cases: A systematic literature review and meta-analysis. J Infect S0163-4453: 30234–30236. [View]
- Long-Quan Li, Huang T, Yong-Qing W, Zheng-Ping W, Yuan Liang et al. (2020) COVID-19 patients’ clinical characteristics, discharge rate, and fatality rate of meta-analysis [published online ahead of print, 2020 Mar 12]. J Med Virol 2020. [View]
- Wang B, Li R, Lu Z, Huang Y (2020) Does comorbidity increase the risk of patients with COVID-19: evidence from meta-analysis. Aging (Albany NY 12: 6049?6057. [View]
- Fu L, Wang B, Yuan T, Chen , Ao Y, et al. (2020) Clinical characteristics of coronavirus disease 2019 (COVID-19) in China: A systematic review and meta-analysis. J Infect 80: 656?665. [View]
- Shi Y, Yu X, Zhao H, Wang H, Zhao R, et al. (2020) Host susceptibility to severe COVID-19 and establishment of a host risk score: findings of 487 cases outside Wuhan. [J] Crit Care 24: 108. [View]
- Huang C, Wang Y, Li X, et al. (2020) Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 395: 497–506. [View]
- Chen N, Zhou M, Dong X, et al. (2020) Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet 395: 507–513. [View]
- Guan WJ, ZY N, Hu Y, et al. (2020) Clinical characteristics of 2019 novel coronavirus infection in China. N Engl J Med 382: 1708–1720. [View]
- Zhou S, Wang Y, Zhu T, et al. (2020) Ct features of coronavirus disease 2019 (COVID-19) pneumonia in 62 patients in Wuhan, China. AJR Am J Roentgenol 2020: 1–8. [View]
- Wang J, Liu JP, Wang YY, et al. (2020) [Dynamic Changes of Chest CT Imaging in Patients With Corona Virus disease-19 (COVID-19)]. Zhejiang Da Xue Xue Bao Yi Xue Ban 49. [View]
- Liao XN, Zhou J, Cao J, et al. (2020) Chest CT features comparison between COVID-19 and a. bacterial pneumonia. Medical Journal of Wuhan university 26.
- Chung M, Bernheim A, Mei X, et al. (2020) CT Imaging Features of 2019 Novel Coronavirus (2019-nCoV). Radiology 295: 202–207. [View]
- Pan Y, Guan H, Zhou S, et al. (2020) Initial CT findings and temporal changes in patients with the novel coronavirus pneumonia (2019-nCoV): a study of 63 patients in Wuhan, China. Eur Radiolv 30: 3306–3309. [View]
- Wu J, Wu X, Zeng W, et al. (2020) Chest CT Findings in Patients With Coronavirus Disease 2019 and Its Relationship With Clinical Features. Invest Radiol 55: 257–261. [View]
- Shi H, Han X, Jiang N, et al. (2020) Radiological findings from 81 patients with COVID-19 pneumonia in Wuhan, China: a descriptive study. Lancet Infect Dis 20: 425–434. [View]
- Yang X, Yu Y, Xu J, et al. (2020) Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: a single-centered, retrospective, observational study. Lancet Respir Med 8: 475–481. [View]
- Zhang MQ, Wang XH, Chen YL, et al. (2020) Zhonghua Jie He He Hu Xi Za Zhi 43: E013.
- Yang K, Ren MH, Xiao YL, et al. (2020) Epidemiological and clinical characteristics of 57 casesof new coronavirus pneumonia in non-epidemic areas. J Third Mil Med Univ 2020.
- Xiang TX, Liu JM, Xu F, et al. (2020) Analysis of clinical characteristics of 49 patientswith novel coronavirus pneumonia in Jiangxi Province. Chinese J Respir Crit Care Med 2020.
- Ma PQ, Yuan YS, Zhang L, et al. (2020) Manifestations of the initial chest CT and its association with laboratory tests in 75 COVID-19 patients. Int J Med Radiol 2020: 1–4.
- Xue HH, Zhang HY, Pang ZH, et al. (2020) Analysis on TCM clinical characteristics of 66 COVID-19 cases in the recover period. Shan J Tradit Chin Med 2020.
- Gong X, Mou FZ, Wei DR, et al. (2020) The clinical characteristics and medication analysis of corona virus disease 2019. World Chinese Medicine 2020.
- Ran J, YP L, QT L, et al. (2020) Study of TCM syndrome in 209 novel coronavirus pneumonia cases of Chongqing in 2020. Journal of Emergency in Traditional Chinese Medicine 2020.
- Yuan J, Sun YL, Zuo YJ, et al. (2020) Clinical characteristics of 223 novel coronavirus pneumonia cases in Chongqing. Journal of Southwest University(Natural Science Edition) 2020.
- Shi J, Yang ZG, Chen Y, et al. (2020) Clinical observation on 49 cases of non-critical coronavirus disease 2019 in Shanghai treated by integrated traditional Chinese and Western medicine. Shanghai Journal of Traditional Chinese Medicine 2020.
- Xiong J, Jiang WL, Zhou Q, et al. (2020) Clinical characteristics, treatment, and prognosis in89 cases of COVID?2019. Medical Journal of Wuhan University(Health Sciences) 2020.
- Chen X, Tong J, Xiang JH, et al. (2020) Retrospective study on the epidemiological characteristics of 139 patients with novel coronavirus pneumonia on the effects of severity. Chongqing Medicine 2020.
- Fang L, Zhu QG, Cheng W, et al. (2020) Retrospective analysis on 308 cases of COVID-19 and clinical application program of Kang Yi Qiang Shen Gong exercise prescription. Shanghai Journal of Traditional Chinese Medicine 2020.
- Cheng KB, Wei M, Shen H, et al. (2020) Clinical characteristics of 463 patients with common and severe type coronavirus disease 2019. Shanghai Medical Journal 2020.
- Chen T, Jiang ZY, Xu W, et al. (2020) Clinical features and CT imaging analysis of 76patiens with corona virus disease 2019. Journal of Jinan University 2020.
- Cheng JL, Huang C, Zhang GJ, et al. (2020) [Epidemiological characteristics of novel coronavirus pneumonia in Henan]. Zhonghua Jie He He Hu Xi Za Zhi 43: E027.
- Han J, Dong XF, Hu F, et al. (2020) Clinical characteristics of 120 patients infected with SARS-CoV-2. Guangdong Medical Journal 2020.
- Xu S, HT H, YG H, et al. (2020) Clinical features of 62 cases of coronavirus disease 2019complicated with acute renal injury. Medical Journal of Wuhan University 2020: 1–5.
- Zhou SY, Wang CT, Zhang W, et al. (2020) Clinical characteristics and treatment effect of 537 cases of novel coronavirus pneumonia in Shandong Province. Journal of Shandong University(Health Sciences) 2020.
- Jin X, Lian JS, Hu JH, et al. (2020) Epidemiological, clinical and virological characteristics of 74 cases of coronavirus-infected disease 2019 (COVID-19) with gastrointestinal symptoms. Gut 69: 1002–1009. [View]
- Song ZY, Yong WX, Li J, et al. (2020) Analysis on TCM syndrome rules of 60 Cases of novel coronavirus pneumonia in Gansu area. Chinese Journal of Information on TCM 2020.
- Dong XC, Li JM, Bai JY, et al. (2020) [Epidemiological characteristics of confirmed COVID-19 cases in Tianjin]. Zhonghua Liu Xing Bing Xue Za Zhi 41: 638–642.
- Sun WW, Ling F, Pan JR, et al. (2020) [Epidemiological characteristics of 2019 novel coronavirus family clustering in Zhejiang Province]. Zhonghua Yu Fang Yi Xue Za Zhi 54: E027.
- Li K, Wu J, Wu F, et al. (2020)The clinical and chest CT features associated with severe and critical COVID-19 pneumonia. Invest Radiol 2020. [Epub ahead of print: 29 Feb 2020].
- Zhou F, Yu T, Du R, et al. (2020) Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet 395: 1054–1062. [View]
- Wu J, Liu J, Zhao X, et al. (2020) Clinical characteristics of imported cases of COVID-19 in Jiangsu Province: a multicenter descriptive study. Clin Infect Dis 2020. [Epub ahead of print: 29 Feb 2020].
- Zhao W, Zhong Z, Xie X, et al. (2020) Relation between chest CT findings and clinical conditions of coronavirus disease (COVID-19) pneumonia: a multicenter study. AJR Am J Roentgenol 2020: 1–6. [View]
- Wang T, Shi L, Chen YY, et al. (2020) Clinical efficacy analysis of 50 cases of corona virus disease 2019 in traditional Chinese medicine. Jilin Journal of Chinese Medicine 2020.
- Fang XW, Mei Q, Yang TJ, et al. (2020) Clinical characteristics and treatment analysis of 79 cases of COVID-19. Chinese Pharmacological Bulletin 2020.
- Wu C, Chen X, Cai Y (2020) Risk Factors Associated With Acute Respiratory Distress Syndrome and Death in Patients With Coronavirus Disease 2019 Pneumonia in Wuhan, China. JAMA Intern Med 2020.
- Wang Z, Chen X, Lu Y, Chen F, Zhang W (2020) Clinical characteristics and therapeutic procedure for four cases with 2019 novel coronavirus pneumonia receiving combined Chinese and Western medicine treatment [published correction appears in Biosci Trends. 2020;14(1): E1]. Biosci Trends 14: 64–68. [View]
- Liu Y, Yang Y, Zhang C, et al. (2020) Clinical and biochemical indexes from 2019-nCoV infected patients linked to viral loads and lung injury. Sci China Life Sci 63: 364–374. [View]
- Du Y, Tu L, Zhu P, et al. (2020) Clinical Features of 85 Fatal Cases of COVID-19 from Wuhan. A Retrospective Observational Study. Am J Respir Crit Care Med 201: 1372–1379. [View]
- Zhang X, Li L, Dai GC, et al. (2020) A preliminary study on the clinical characteristicsandchinese medical syndrome of 42 cases of COVID-19 in Nanjing. Journal of Nanjing University of Traditional Chinese Medicine 2020.
- Mo P, Xing Y, Xiao Y (2020) Clinical characteristics of refractory COVID-19 pneumonia in Wuhan, China. Clin Infect Dis 2020.
- Peng YD, Meng K, Guan HQ (2020) [Clinical characteristics and outcomes of 112 cardiovascular disease patients infected by 2019-nCoV]. Zhonghua Xin Xue Guan Bing Za Zhi 48: E004.
- Tian S, Hu N, Lou J (2020) Characteristics of COVID-19 infection in Beijing. [J] J Infect 80: 401–406. [View]
- Wang Z, Yang B, Li Q (2020) Clinical Features of 69 Cases with Coronavirus Disease 2019 in Wuhan, China. Clin Infect Dis 2020.
- Xiong Y, Sun D, Liu Y, et al. (2020) Clinical and high-resolution CT features of the COVID- 19 infection: comparison of the initial and follow-up changes. Invest Radiol 2020. [Epub ahead of print: 03 Mar 2020].
- Li YY, Wang WN, Lei Y, et al. (2020) [Comparison of the clinical characteristics between RNA positive and negative patients clinically diagnosed with 2019 novel coronavirus pneumonia]. Zhonghua Jie He He Hu Xi Za Zhi 43: E023.
- Xu X, Yu C, Qu J, et al. (2020) Imaging and clinical features of patients with 2019 novel coronavirus SARS-CoV-2. Eur J Nucl Med Mol Imaging 47: 1275–1280.
- Xu Y-H, Dong J-H, An W-M, et al. (2020) Clinical and computed tomographic imaging features of novel coronavirus pneumonia caused by SARS-CoV-2. J Infect 80: 394–400. [View]
- Xiao KF, Shui LL, Pang XH, et al. (2020) The clinical features of the 143 patients withCOVID-19 in north-east of Chongqing. Journal of Third Military Medical University 2020.
- Sun HY, YF B, Zhu ZG, et al. (2020) A preliminary study on TCM syndrome characteristics of 88 patients with COVID-19 in Tianjin. J Tradit Chin Med 2020.
- Xu B, Fan CY, Zou YL, et al. (2020) Analysis of traditional Chinese medicine syndromes of 46 cases of COVID-19. Chinese journal of experimental traditional medical formulae 2020 Wan S, Xiang Y, Fang W, et al. (2020) Clinical features and treatment of COVID-19 patients in northeast Chongqing. J Med Virol 92: 797–806. [View]
- Yang W, Cao Q, Qin L, et al. (2020) Clinical characteristics and imaging manifestations of the 2019 novel coronavirus disease (COVID-19): A multi-center study in Wenzhou city, Zhejiang, China. J Infect 80: 388–393.
- Xu XW, Wu XX, Jiang X-G, et al. (2020) Clinical findings in a group of patients infected with the 2019 novel coronavirus (SARS-Cov-2) outside of Wuhan, China: retrospective case series. BMJ 368: 606.
- Zhang JJ, Dong X, Cao YY, et al. (2020) Clinical characteristics of 140 patients infected with SARS-CoV-2 in Wuhan, China. Allergy 2020. [Epub ahead of print: 19 Feb 2020].
- Wang D, Hu B, Hu C, et al. (2020) Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA 323: 1061.
- Liu K, Fang YY, Deng Y, et al. (2020) Clinical characteristics of novel coronavirus cases in tertiary hospitals in Hubei Province. Chin Med J (Engl) 133: 1025–1031. [View]
- Ahn DG, Shin HJ, Kim MH, et al. (2020) Current Status of Epidemiology, Diagnosis, Therapeutics, and Vaccines for Novel Coronavirus Disease 2019 (COVID-19). J Microbiol Biotechnol 30: 313–324. [View]
- Hong KH, Lee SW, Kim TS, et al. (2020) Guidelines for Laboratory Diagnosis of Coronavirus Disease 2019 (COVID-19) in Korea. Ann Lab Med 40: 351–360. [View]
- Li X, Geng M, Peng Y, Meng L, Lu S (2020) Molecular immune pathogenesis and diagnosis of COVID-19. J Pharm Anal 10: 102–108. [View]
- Nicola M, O’Neill N, Sohrabi C, Khan M, Agha M, Agha R. (2020) Evidence based management guideline for the COVID-19 pandemic - Review article. Int J Surg 77: 206–216. [View]
- Kokkinakis I, Selby K, Favrat B, Genton B, Cornuz J (2020) Performance du frottis nasopharyngé-PCR pour le diagnostic du Covid-19 - Recommandations pratiques sur la base des premières données scientifiques [Covid-19 diagnosis: clinical recommendations and performance of nasopharyngeal swab-PCR]. Rev Med Suisse 16: 699–701. [View]
Article Type
Research Article
Publication history
Received: July 08, 2020
Accepted: July 15, 2020
Published: July 18, 2020
Citation:
GORGUN S (2020) A Comprehensive COVID-19 Meta-Analysis: Clinical Data of 18,450 Patients. Clar J Infect Dis Ther 01(01): 13–23.
Selim GORGUN*
Department of Microbiology and Clinical Microbiology Laboratory, Training and Research Hospital, Health Sciences University Turkey, Ilkad?m, Samsun, Turkey
*Corresponding author
Selim GORGUN,
Department of Microbiology and Clinical Microbiology Laboratory,
Training and Research Hospital,
Health Sciences University Turkey,
Ilkad?m, Samsun,
Turkey;